CAPA & Deviations
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreA systematic method for identifying the underlying cause of a quality issue — not the symptom — so corrective action can actually prevent recurrence.
Inspectors distinguish between fixing a symptom and addressing a cause. A weak root cause like 'human error' almost always signals insufficient investigation and weak CAPA evidence.

Root Cause Analysis (RCA) is a structured investigation method used to identify the fundamental underlying cause of a quality event, rather than its visible symptom or proximate trigger.
The distinction matters: a symptom is what you see (a failed batch), a proximate cause is what directly preceded it (wrong temperature), and the root cause is what allowed it to happen (a missing alarm in the control system design). Corrective actions targeting symptoms fail. Actions addressing root causes prevent recurrence.
"Human error" written as a root cause is one of FDA's most-cited inspection findings. It indicates the investigator stopped at the last person who touched the process rather than asking why a human-dependent step existed at all, why training did not prevent the error, or why a control check did not detect it.
Strong RCA evidence shows the investigator asked "why" multiple times, considered system-level causes, and documented the logic chain. This becomes the foundation for credible CAPAs.
If your root cause is 'operator made a mistake', you have almost certainly not completed your investigation. The real question is: why did the system allow a single operator mistake to cause a quality event?
RCA is implicit in most GxP regulations and explicit in modern quality frameworks:
Several analytical tools support root cause investigation. Use the one fitted to the event's complexity:
Quality systems with strong RCA practice share:
Complere provides a controlled root cause framework inside CAPA and Deviation records, preventing shallow single-word causes and enforcing evidence-backed conclusions.
Common questions about Root Cause Analysis (RCA) sourced from regulatory references and inspection patterns.
Root cause analysis is the structured investigation that identifies the underlying reason a deviation, complaint, or audit finding occurred — not just the surface symptom. It's the foundation of every CAPA, deviation closure, and recurring-issue prevention program.
Inspectors expect a method appropriate to the issue's complexity: 5 Whys for simple deviations, Fishbone (Ishikawa) for multi-cause issues, FMEA for risk-prioritized failure modes, and fault tree analysis for safety-critical events. Method choice should be documented in the SOP.
Until removing the identified cause would prevent recurrence — not just until a contributing factor is found. Premature closure on operator error or 'human factors' is a common 483 finding; inspectors expect investigation of why the system allowed the error.
Two patterns drive most findings: closure at the first plausible cause without testing alternatives, and corrective actions disproportionate to the cause (e.g., retraining when the SOP itself was unclear). Both signal weak investigation discipline to inspectors.
Complere captures the investigation method, evidence reviewed, ruled-out hypotheses, and confirmed root cause as linked records — so inspectors see the analytical path, not just the conclusion, and recurring causes surface across deviations and CAPAs over time.
Explore related topics, modules, and compliance resources for a deeper understanding of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore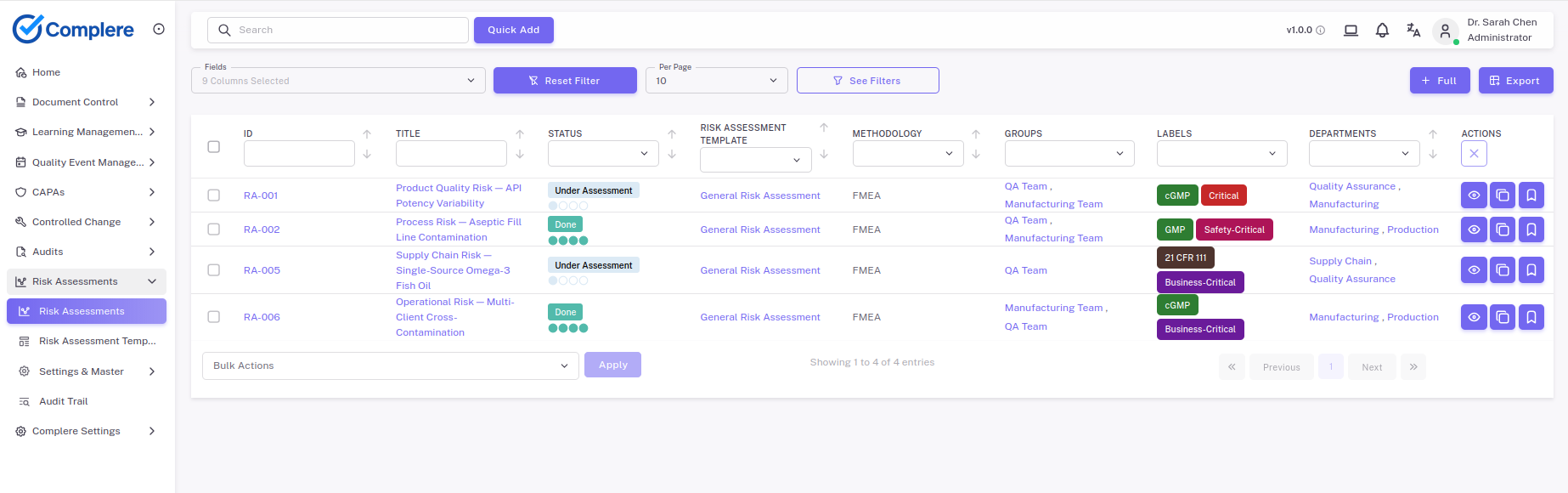
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreExplore how Complere's controlled RCA taxonomy and guided investigation templates drive deeper root causes and stronger CAPA evidence.