Keep one approved source of truth
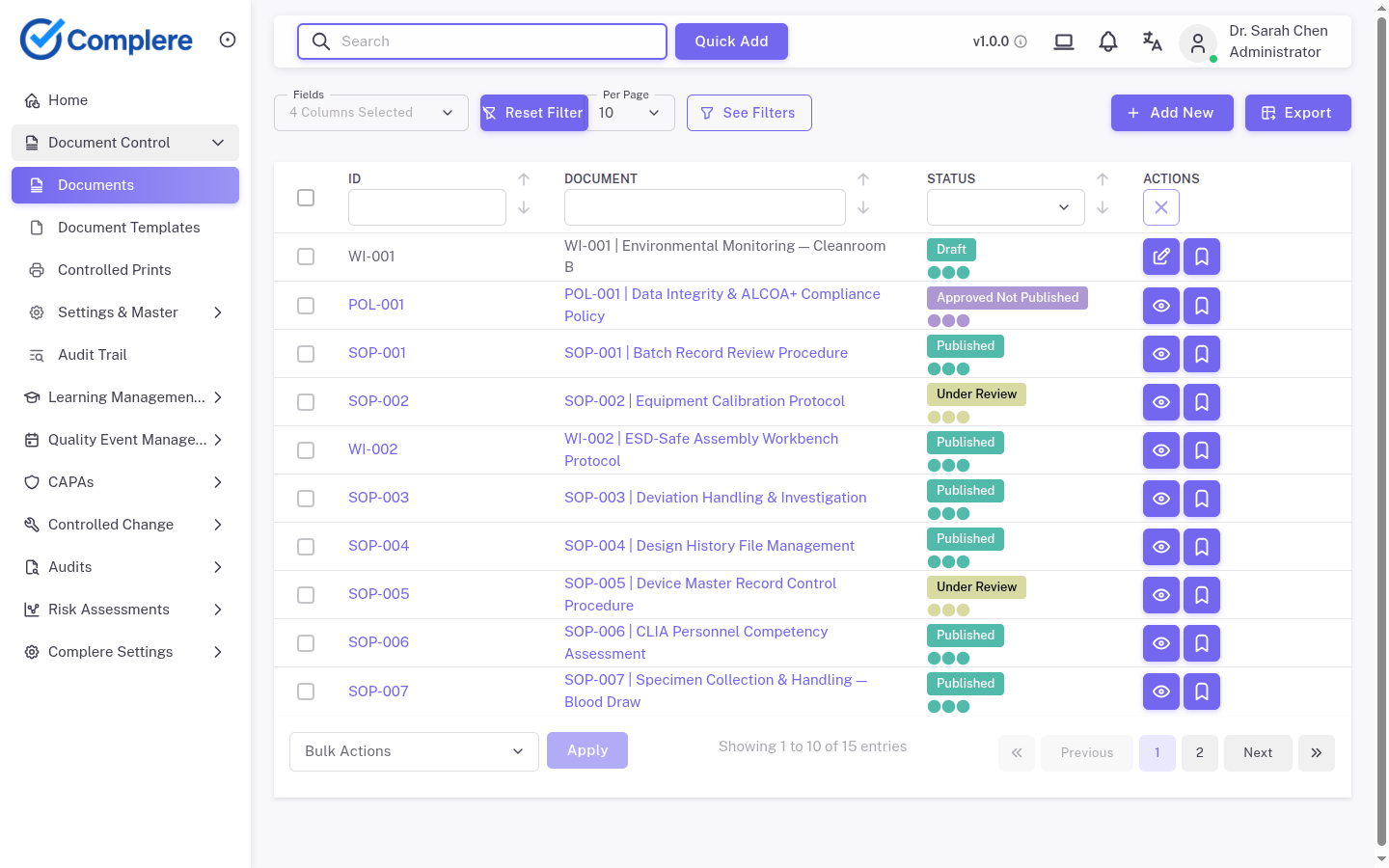
Draft, review, approve, retire, and archive documents with clear revision history.
Move document lifecycles into one controlled environment with versioning, review workflows, controlled print visibility, and connected training triggers. Under audit, teams show the effective SOP, prior revisions, approval signatures, and training completion tied to each release — the same evidence pattern regulators expect for GxP document control.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Draft, review, approve, retire, and archive documents with clear revision history.
Tie revised procedures to training assignments and completion evidence.
Retrieve current, obsolete, and printed document evidence from one place.
Auditors expect a clear path from draft to retirement — with role-based approvals, electronic traceability, and controlled distribution at every state.
Authors work in a controlled workspace with ownership, metadata, and change history captured from the first revision.
Parallel or sequential review with comments resolved before approval; every reviewer action is time-stamped.
Electronic signatures from authorized approvers; no effective date until the approval chain is complete.
Only the current effective version is distributed for use; controlled copies and downloads are logged.
When a new revision is effective, prior versions move to superseded status but remain readable for audit and batch release context.
Obsolete documents are retained per retention policy with read-only access and full audit trail for inspections.
Complere eQMS is not just a repository — you create, edit, and regenerate controlled documents in place, in the Microsoft Office tools your team already uses, without breaking version control or the audit trail.
Click Live Edit on a controlled document and it opens directly in your installed desktop Microsoft Word, Excel, or PowerPoint. When you save, the file streams straight back into the eQMS as a new controlled version — no download, re-upload, or copy-paste.
Live Edit runs on a standards-based WebDAV server with real document locking — a second user cannot edit while someone else holds the lock, and admins can force-release it. Every save becomes a controlled new version with a 21 CFR Part 11 audit entry and a stakeholder notification.
Reviewers open any controlled PDF or Microsoft Office document — any prior version or attached file — in the browser, with no local application required, before they review or sign.
Templates carry merge-field placeholders that Complere auto-detects on upload and auto-fills with live values on generate, regenerate, or print — with a live preview. Regeneration re-applies current values without altering approval status or version history.
Compare any two versions side by side, with field-level old-versus-new values for every change event — the difference view reviewers and auditors need without reading two documents in full.
An AI assistant can summarize a controlled document and produce a redline-style diff between versions to speed review. AI output is clearly labelled, advisory only, and never written to the controlled record — the validated audit trail stays the single source of truth.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Configure review and approval pathways by document type, site, or function.
Show which copies were issued, when, and under which status.
Use default fields, ownership, and taxonomy to simplify retrieval.
Keep impact analysis visible across the quality system.
Document editing, collaboration, and Part 11 control in Complere eQMS.
Yes. Complere eQMS includes Live Edit — click Live Edit on a controlled document and it opens directly in your installed desktop Microsoft Word, Excel, or PowerPoint application. When you save, the file streams straight back into the eQMS as a new controlled version, with no download, re-upload, or copy-paste.
Yes. Every saved live edit is captured as a controlled new version with a 21 CFR Part 11 audit-trail entry and a stakeholder notification. Live editing runs on a standards-based WebDAV server with real document locking, so a second user cannot acquire an edit lock while someone else is editing, and administrators can force-release a stuck lock.
Yes. An in-browser viewer renders controlled PDF and Microsoft Office documents — any prior version or attached file — directly in the browser, with no local application required.
Yes. Templates can contain merge-field placeholders that Complere auto-detects when a Word file is uploaded and auto-fills with live values when a document is generated, regenerated, or printed, with a live preview before you commit.
Yes. Complere shows a clear before-and-after difference between document versions, including field-level old-versus-new values for every change event, so reviewers and auditors can see exactly what changed.
Use document control as the starting module for digital rollout, then expand into CAPA, audits, change, and training.