§11.10 record controls built in
System access controls, audit trails, record retention, and authority checks are implemented at the platform level — not configured separately per workflow.
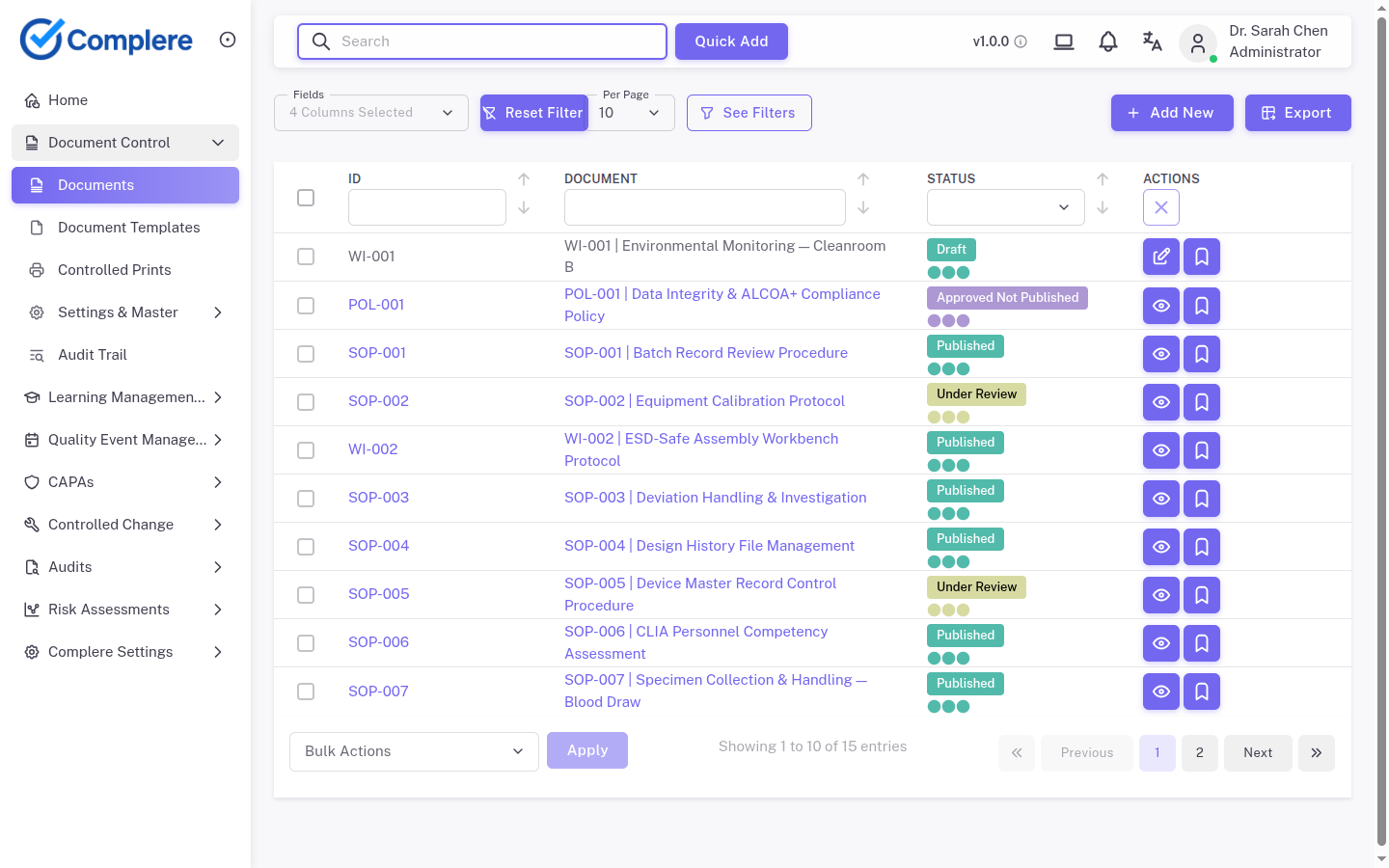
FDA 21 CFR Part 11 establishes the technical and procedural controls required for electronic records and electronic signatures to be considered trustworthy equivalents to paper records. Complere implements the §11.10 and §11.50 requirements across every quality workflow — from document approvals and CAPA sign-off to training records and audit findings — so regulated teams can replace wet-ink signatures with confidence.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
System access controls, audit trails, record retention, and authority checks are implemented at the platform level — not configured separately per workflow.
Each signature captures the action type (approve, reject, review), the user's intent statement, and the record context — meeting the §11.50 manifestation requirement.
Complere addresses Annex 11 requirements for access control, audit trail, change control, backup, and business continuity relevant to regulated European operations.

Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
Unique user identity confirmed via password, SSO, or MFA before any quality action is permitted.
A governed quality step requires signature — document approval, CAPA closure, training confirmation.
User re-confirms identity, selects the meaning of the action, and the system timestamps and links the signature to the record.
The signed record and its signature context are protected from alteration and can be retrieved for inspection at any time.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Complere's CSV programme ensures the system operates accurately and consistently throughout its validated lifetime.
Role-based access controls, session management, and user provisioning workflows restrict system entry to individuals with defined responsibilities.
Independent, tamper-evident audit trails are generated automatically — users cannot edit or delete trail entries.
Every electronic signature displays the signer's name, the date and time of signing, and the meaning of the signature (approval, review, or other action).
Comparing eQMS vendors on Part 11? See Best eQMS for 21 CFR Part 11 — 6 platforms scored on §11.10 / §11.50 / §11.70 / §11.100 with ATS evidence.
Our team walks quality leads and compliance officers through the specific controls, configuration options, and validation evidence that satisfy FDA and EU GMP electronic records requirements.