Competency evidence is critical
Training, qualifications, and recurring assessment visibility are constant operational needs.
Clinical, diagnostic, and laboratory quality teams need governance that fits CLIA/credentialing and GxP-adjacent expectations: controlled SOPs with draft–review–approve–effective lifecycle, competency evidence, investigations, and management review — without turning the lab into an IT project. See document control for how procedure lifecycle and approvals stay inspection-ready.

The quality and compliance pressures unique to your regulated market — and how Complere is built to address them.
Training, qualifications, and recurring assessment visibility are constant operational needs.
Deviations, complaints, and corrective actions benefit from more formal workflows and dashboards.
Leaders need recurring visibility into overdue work, trends, and compliance status.
The regulatory frameworks, guidance documents, and audit expectations that govern quality operations in this sector.
Clinical Laboratory Improvement Amendments • Personnel qualifications • Proficiency testing • Quality control
College of American Pathologists inspection standards • Checklist-based accreditation • Peer inspection readiness
Medical laboratory quality and competence • Pre-analytical and post-analytical process control • Method validation
The Complere modules most commonly used by teams in this industry to manage compliance and quality operations.
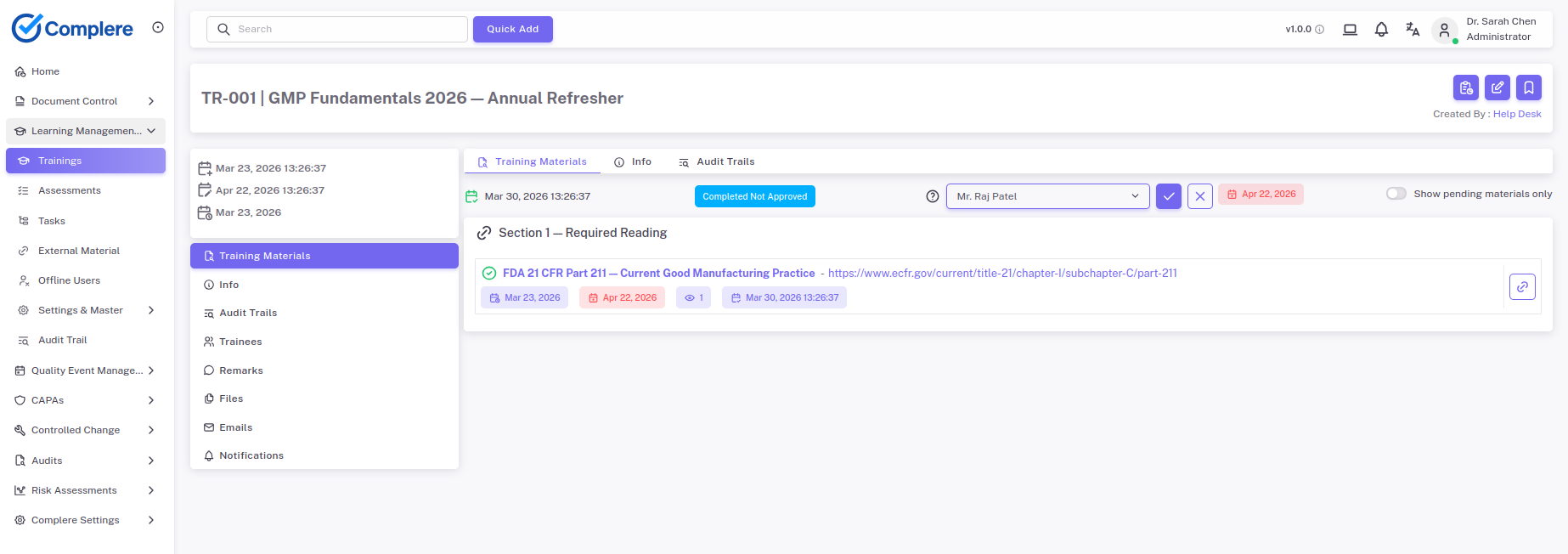
Assign role-specific competency curricula to lab staff, document assessments and re-qualification events, and produce training records that satisfy CLIA and ISO 15189 personnel qualification requirements.
Explore training & competencyLog test failures, equipment issues, and process deviations with structured investigation fields — so root cause analysis and corrective actions follow a consistent, audit-visible workflow across all lab quality events.
Explore CAPA & deviationsSurface open CAPAs, overdue training, and audit follow-up items in real-time dashboard views — giving lab managers and quality leads immediate visibility into quality system health without manual reporting.
Explore dashboards & KPIsExplore related modules, compliance topics, and guides to build a complete picture of your quality system.
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore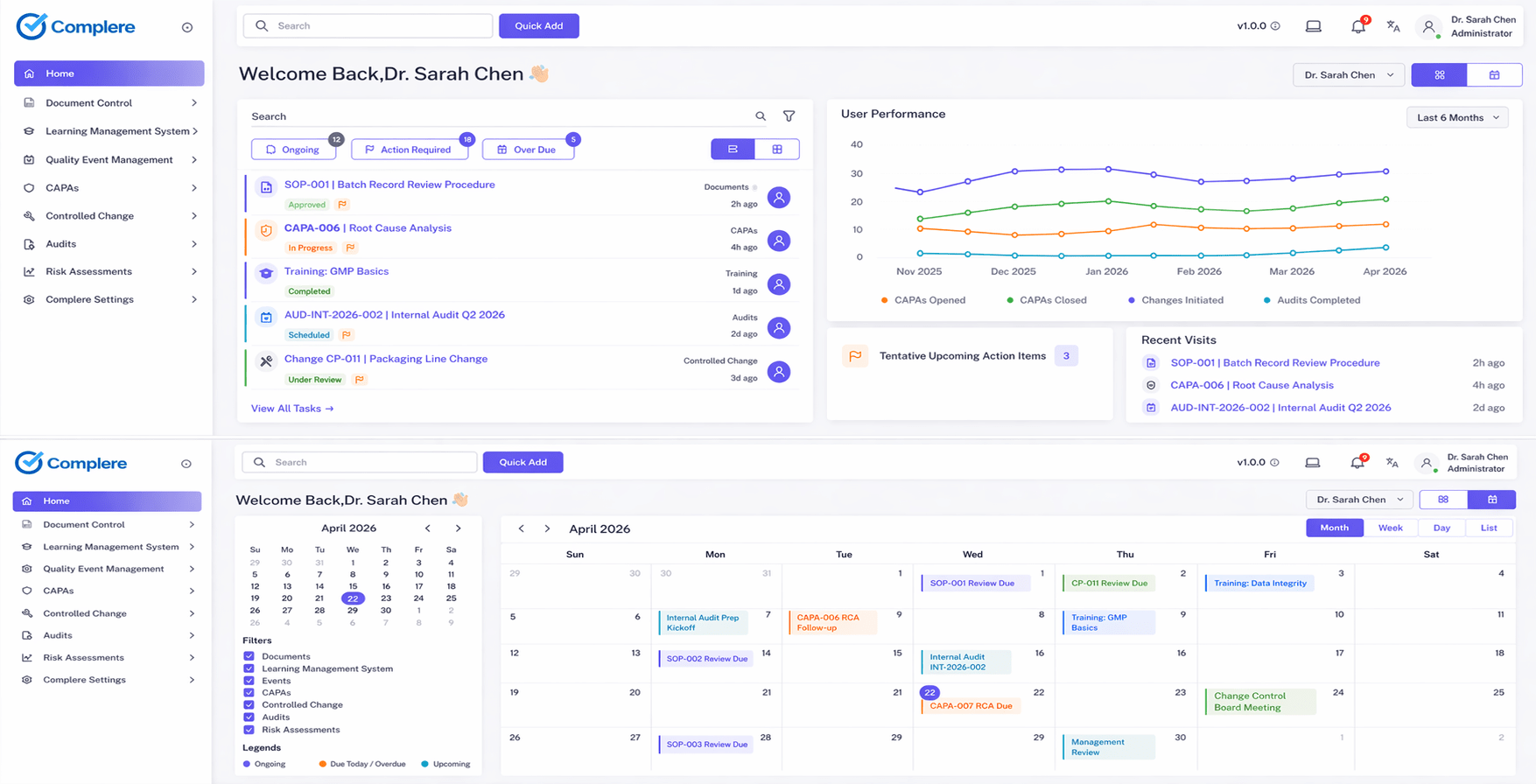
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore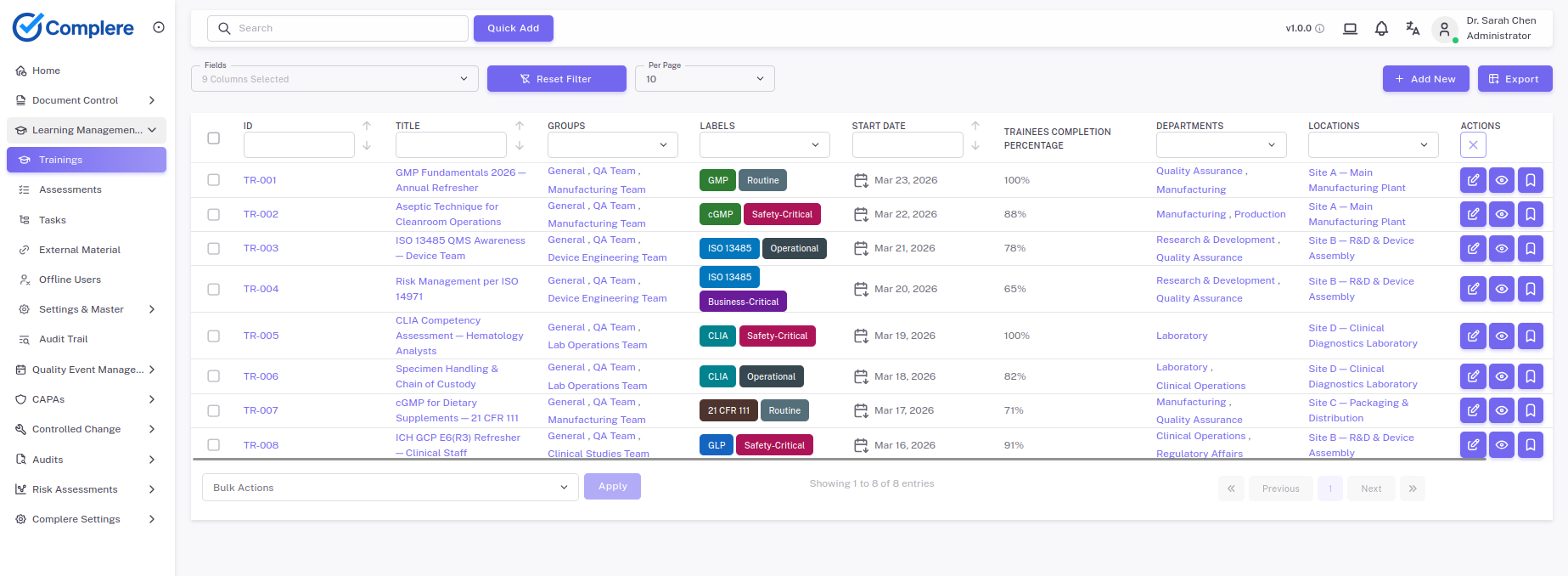
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreFrom training and competency management to audit follow-up and leadership visibility, Complere adapts to your environment.