Score risks across quality events
Use severity, probability, and detectability matrices to evaluate risk consistently across products, processes, and quality events.
Risk management in regulated environments requires evidence that risks are identified, assessed, mitigated, and reviewed — not just documented in a spreadsheet. Complere structures risk assessments with scored matrices, mitigation tracking, and residual risk sign-off, all linked to related CAPA actions, change records, and audit findings — so risk decisions are traceable and inspection-ready.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Use severity, probability, and detectability matrices to evaluate risk consistently across products, processes, and quality events.
Connect identified risks to corrective actions, change decisions, and audit findings — so inspectors see the full risk-to-resolution story in one place.
Track mitigation effectiveness, residual risk acceptance, and scheduled reassessment with signed, auditable records.
Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
Risk assessments in Complere follow a structured, traceable process: risk identification, scored assessment, mitigation planning, implementation, and residual risk sign-off — all linked to the CAPA actions, change records, and audit findings that arise from or inform the risk. ICH Q9 and ISO 14971 obligations are addressed through consistent scoring criteria and periodic review cycles with evidence of completion.
Capture the risk source, affected product or process, and potential impact scope.
Apply severity, probability, and detectability ratings to generate a risk priority score. Structured criteria ensure consistent evaluation across teams and sites.
Assign corrective or preventive actions and link directly to CAPA or change records so follow-through is visible and owned.
Confirm residual risk is acceptable with electronic sign-off. Schedule periodic reassessment so reviews are documented and overdue risks are visible.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Structure scoring criteria by severity, probability, and detectability. Consistent thresholds ensure risk levels are comparable across assessments.
Trace risks to related corrective actions, change decisions, and audit findings — all accessible from within the risk record without switching modules.
Require electronic approval for residual risk before closure — with reason captured and timestamped in the audit trail.
Support pharmaceutical and medical device risk management obligations with structured, audit-ready evidence — not a spreadsheet workaround.
Risk management frameworks addressed by this module.
The international guideline for quality risk management in pharmaceutical development and manufacturing — covering risk assessment, communication, and review throughout the product lifecycle.
The international standard for application of risk management to medical devices — requiring identification, evaluation, control, and monitoring of risks associated with device safety.
Electronic records and signature requirements that apply to risk assessment records created, modified, maintained, archived, and transmitted in electronic form.
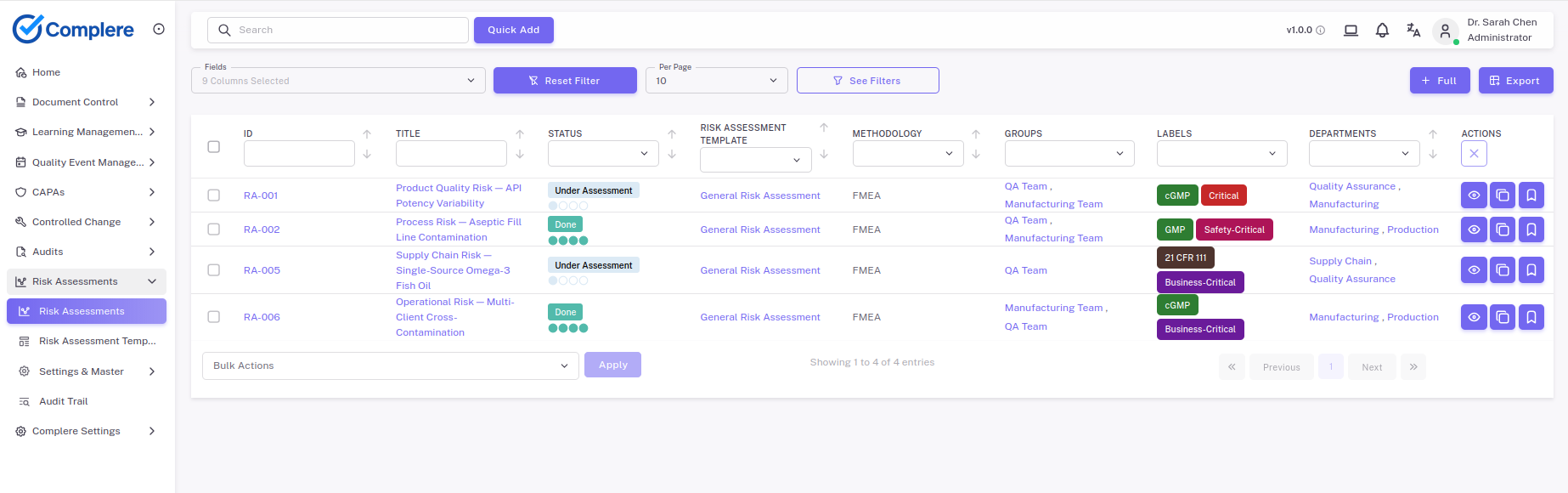
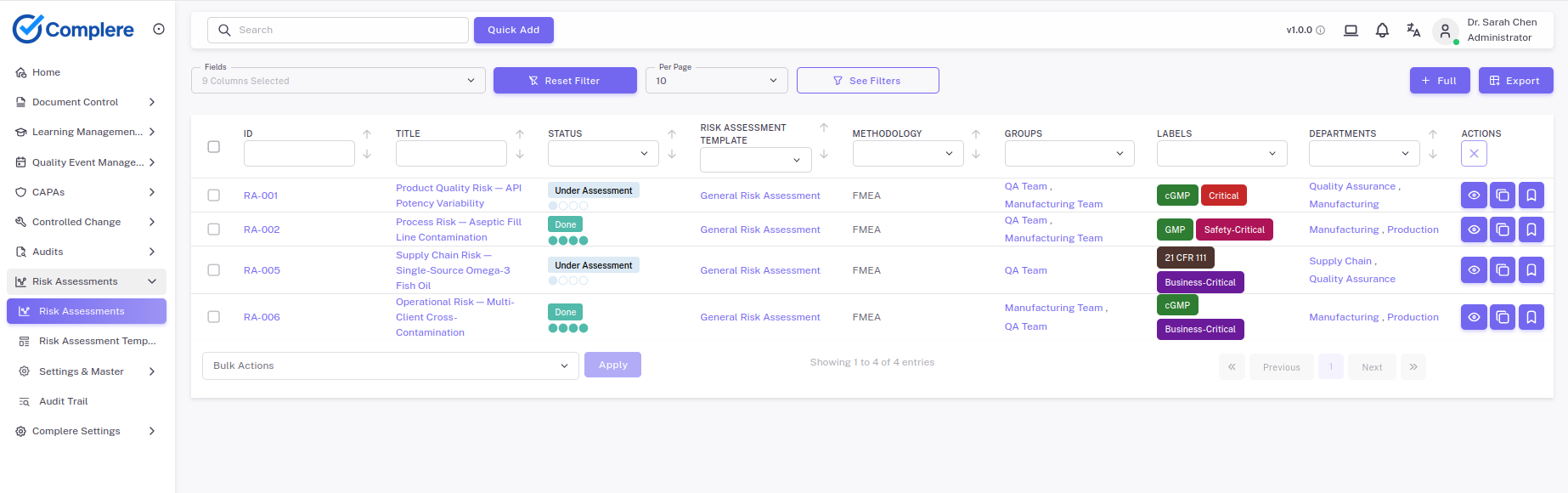
Risk assessments in Complere are linked to the quality events, CAPA actions, and change records that drive them — so risk decisions are traceable from identification to closure.