Quality work spans multiple organizations
Records and actions often involve both internal teams and customer-facing commitments.
Contract manufacturers and CDMOs need multi-site and multi-customer governance: shared standards where they add leverage, and clear separation where client or product context differs. Complere supports audit management, deviations, change control, and training evidence across sites so planning, findings, corrective actions, and closure stay visible in one governed flow.

The quality and compliance pressures unique to your regulated market — and how Complere is built to address them.
Records and actions often involve both internal teams and customer-facing commitments.
The quality system must support defensible answers about documents, deviations, and corrective actions.
Leaders need comparable status across programs and customers.
The regulatory frameworks, guidance documents, and audit expectations that govern quality operations in this sector.
The FDA electronic records and signatures regulation — governing audit trail, access controls, and signature requirements for digital quality systems used in GMP-regulated contract manufacturing environments.
The EU GMP computerized systems guideline — covering validation, audit trails, data integrity, access controls, and supplier oversight for computerized quality systems in contract manufacturing.
The ICH pharmaceutical quality system guideline — establishing quality management requirements for CMOs including change management, CAPA, management review, and continual improvement across the product lifecycle.
The Complere modules most commonly used by teams in this industry to manage compliance and quality operations.
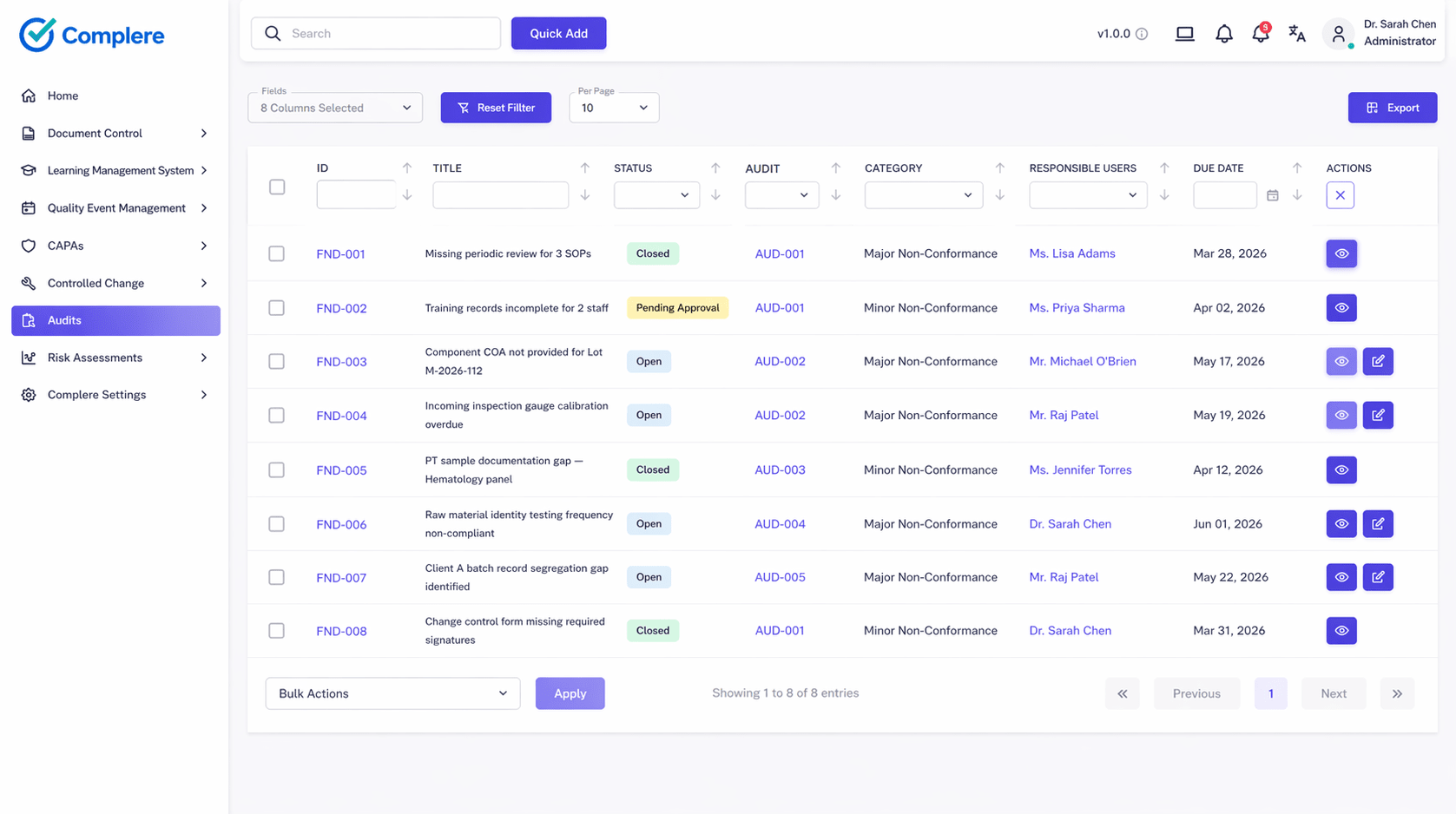
Manage customer-facing audits, internal GMP audits, and supplier qualification programs in one workflow — with checklists, findings, and response evidence accessible to both internal QA and client auditors.
Explore audit managementCapture deviations from customer SOPs or batch specifications and route through structured root cause and CAPA workflows — producing audit-ready closure records that satisfy client quality agreements and EU GMP Annex 11 expectations.
Explore CAPA & deviationsTrack open CAPAs, audit finding closure, and client-specific quality KPIs across multiple programs in one dashboard — giving quality leadership cross-account visibility without switching between spreadsheets.
Explore dashboards & KPIsMaintain inspection-ready evidence across all customer programs — with linked documents, training records, batch records, and change history retrievable by product, client, or audit reference number.
Explore audit confidenceExplore related modules, compliance topics, and guides to build a complete picture of your quality system.
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreComparing eQMS vendors? See Best eQMS for CDMOs and CMOs — 6 platforms scored on multi-client tagging, change impact, and client-audit readiness.
Our demos cover multi-site quality governance, customer-facing evidence, deviation management, and audit follow-up in contract manufacturing environments.