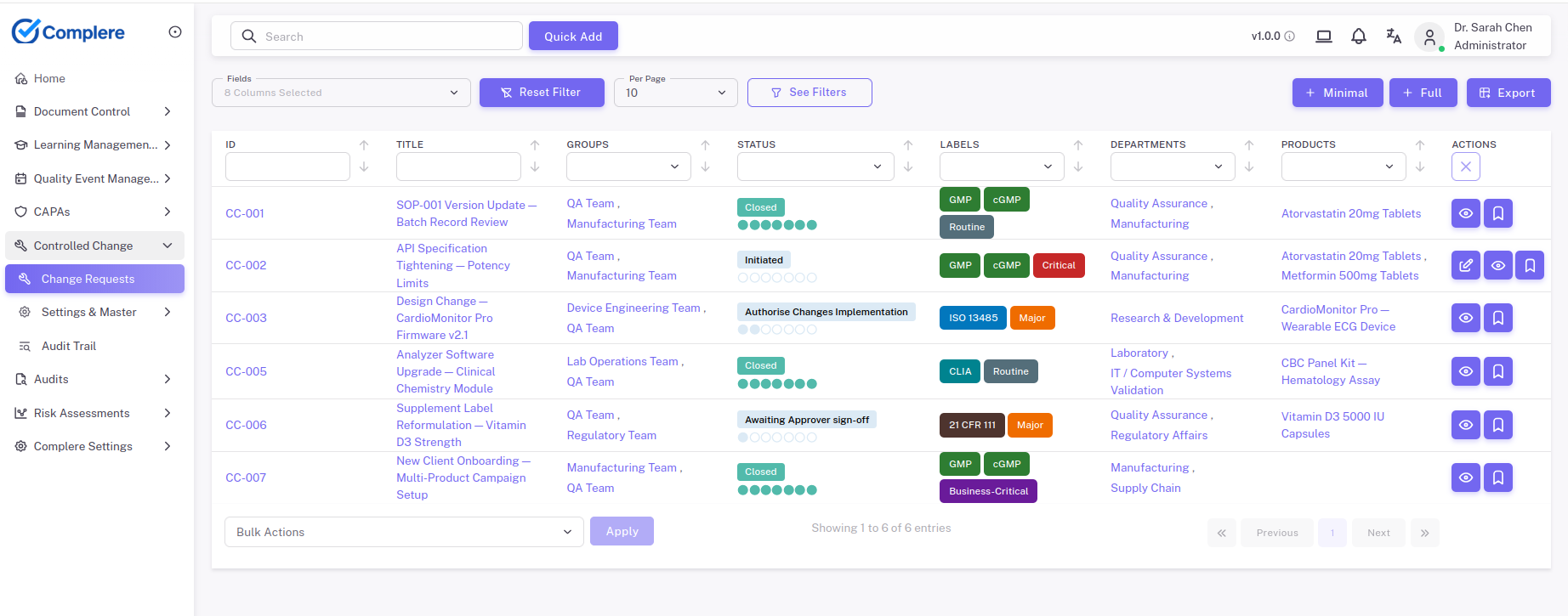
See impact before execution
Use structured review prompts for procedures, products, systems, and training.
Structure changes so teams can evaluate product, process, documentation, training, and validation impact before execution, then show what was completed and verified after release.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Use structured review prompts for procedures, products, systems, and training.
Bring reviewers, implementers, due dates, and evidence requests into one workflow.
Tie changes to required testing, documentation updates, and competency actions.
Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
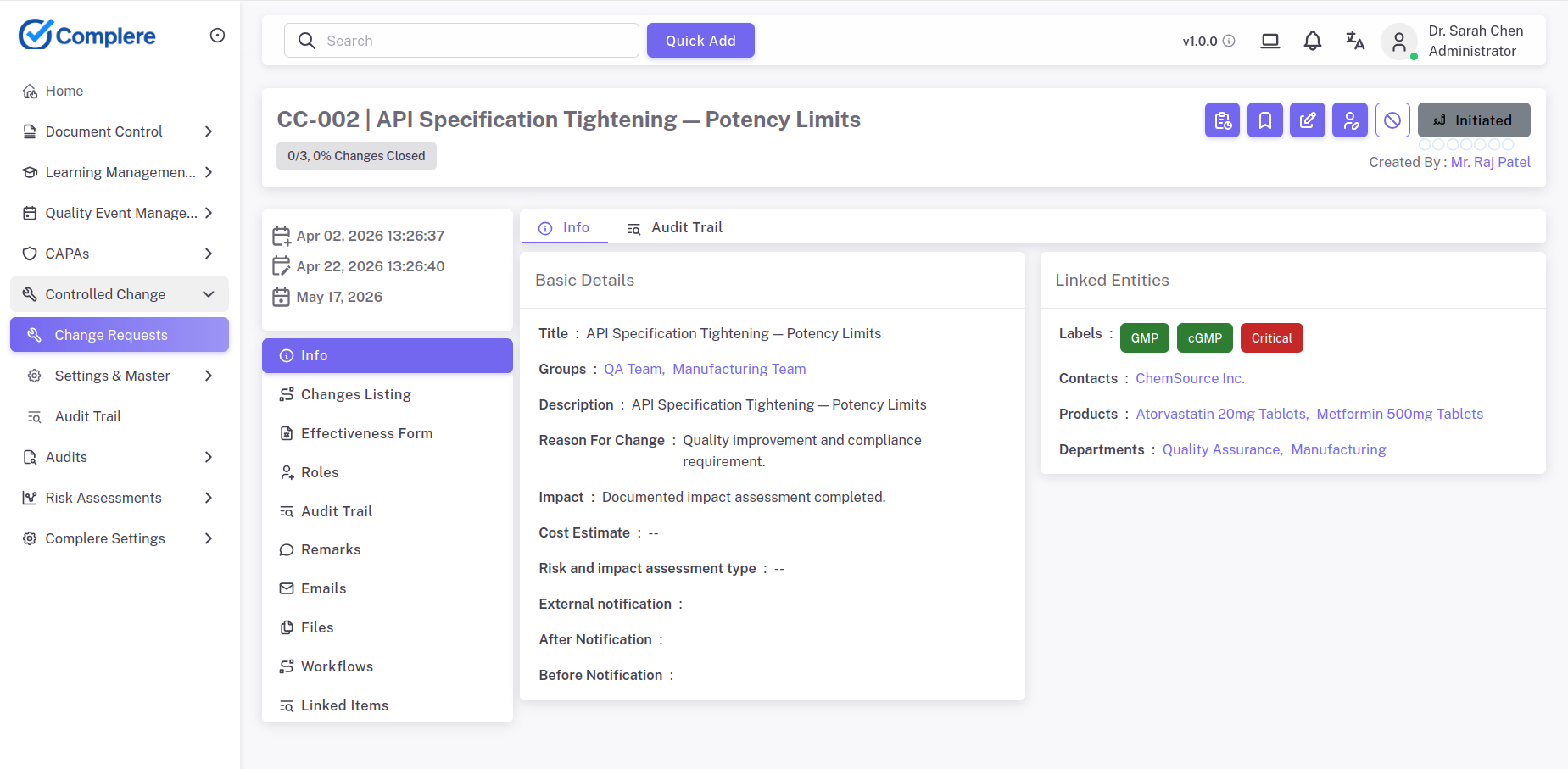
Change control in Complere is a controlled, traceable process: impact assessment, approvals, implementation tasks, linked document updates, triggered training, and closure evidence — so inspectors can follow the full history from request to verified release. Where changes touch manufacturing execution, lab (LIMS), ERP material master, or HR training data, impact and interfaces can be documented in the record so cross-system alignment is explicit — alongside your SSO and integration standards.
Capture the proposed change, rationale, and scope.
Structured prompts for product, process, facility, IT, documentation, training, risk, and validation — so linked SOP updates and retraining are identified before go-live, not discovered in an audit.
Track actions, dependencies, and required updates before release.
Confirm the change is complete and controlled before closure.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Integration landscape is documented per change: typical named systems include ERP (material/status), LIMS / lab data, MES or batch execution, HRMS for org data, and SSO for identity — with impact on Complere records, training, and validation noted before release.
Keep the sequence of review, approval, and release visible.
Show who needs to update procedures or complete retraining before go-live.
Confirm execution with attachments, comments, and sign-off checkpoints.
Use related records to explain why a change was raised and how it was closed.
Change control connects change decisions to their downstream effects — document updates, training actions, and validation evidence — in one visible workflow.