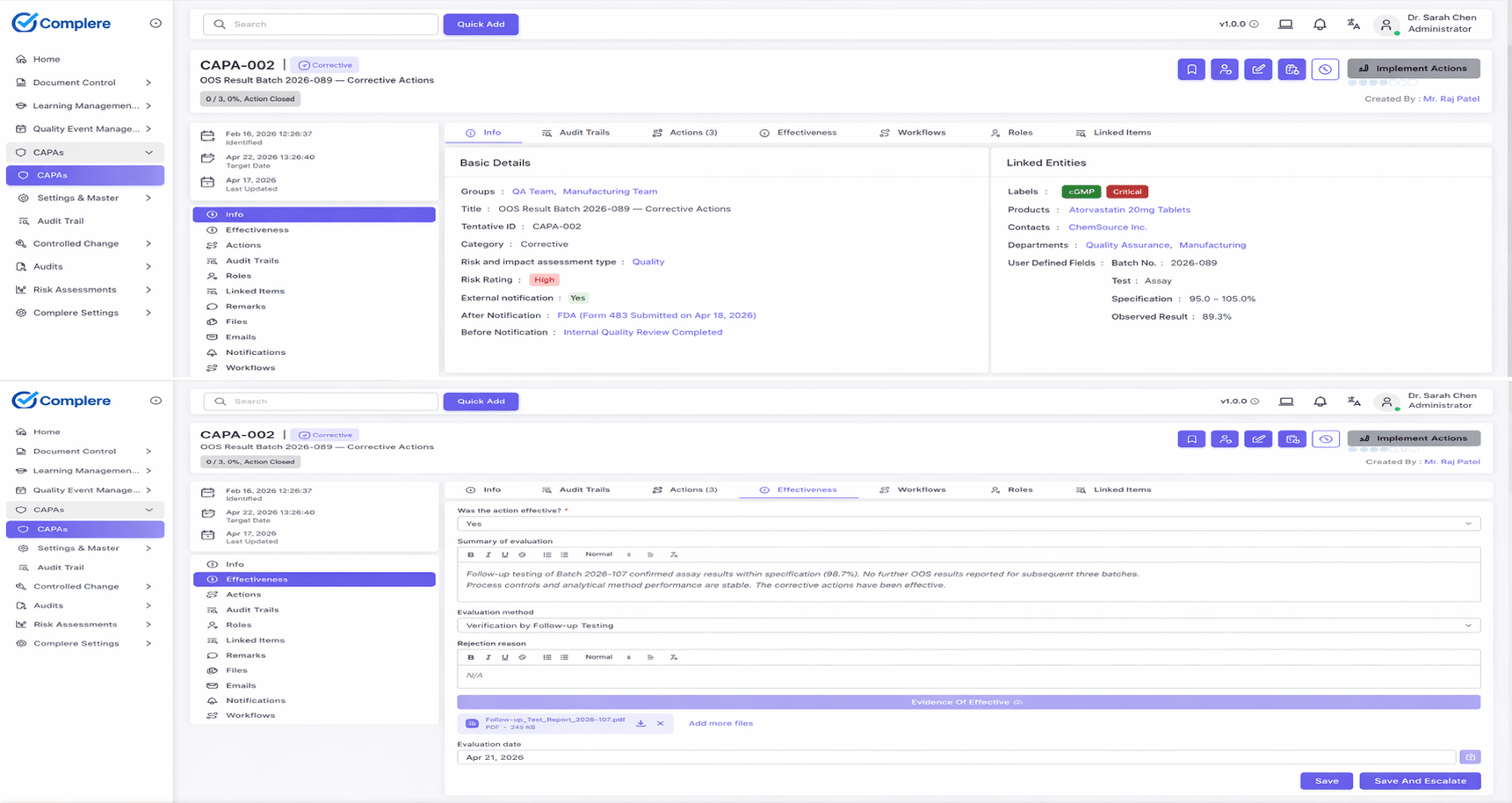
Reduce CAPA cycle time
Make owners, due dates, escalations, and related records visible from the start.
Bring deviations, nonconformances, and complaints into a consistent process with risk-based prioritization, cross-functional action plans, and evidence-backed closure.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Make owners, due dates, escalations, and related records visible from the start.
Investigation notes, related events, and supporting evidence stay linked to the action plan.
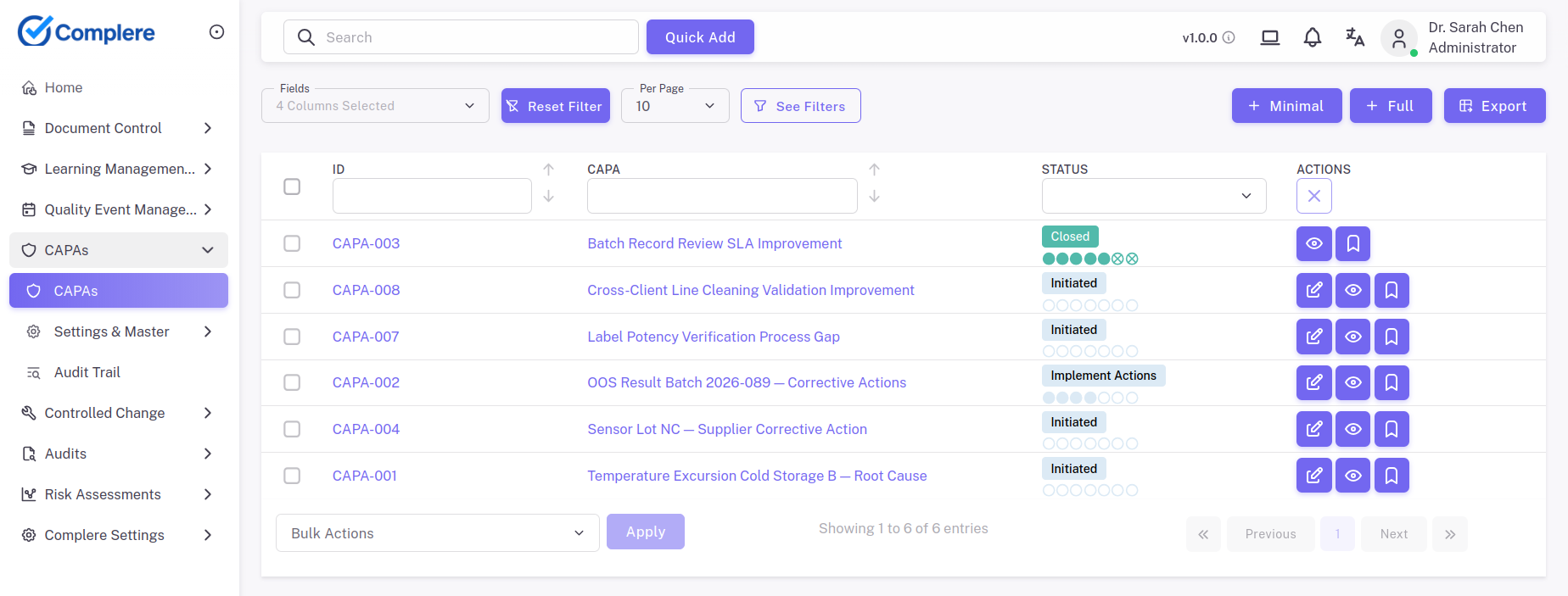
Dashboards expose overdue work, repeat issues, and bottlenecks before they become inspection findings.
Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
End-to-end quality event handling: from issue logging and investigation through root cause analysis (including tools such as 5 Whys or fishbone where your SOP requires them), assigned corrective and preventive actions with owners and due dates, to effectiveness checks and formal closure — with every transition visible to auditors.
Capture the event, initial impact, and containment actions.
Route ownership and collect evidence, interviews, and analysis notes.
Define corrective and preventive actions with due dates and dependencies.
Close only when evidence shows recurrence risk has been reduced.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Standardize intake fields, categories, severity, and containment actions — every save is attributed under FDA 21 CFR Part 11–ready controls with EU GMP Annex 11–aligned audit trail and access rules.
Track analysis, corrective actions, preventive actions, and verification checkpoints.
Tie issues to upstream causes and downstream remediation evidence.
Approvals use Part 11–ready electronic signatures with identity controls; every view and edit is attributed under configurable role-based access — consistent with Annex 11 expectations for computerized systems.
CAPA and deviation workflows become stronger when they share documents, audit findings, risk signals, and management review reporting.