Quality systems mature quickly under pressure
Teams outgrow spreadsheets and manual binders as product lines and partners expand.
Nutraceutical and supplement manufacturers face growing regulatory pressure — from GMP quality controls and batch traceability to labelling compliance and supplier oversight. Complere helps teams standardize documents, deviations, training, and supplier quality with evidence that scales as rules tighten — including CAPA and deviations with end-to-end handling, owners, due dates, and effectiveness checks.

The quality and compliance pressures unique to your regulated market — and how Complere is built to address them.
Teams outgrow spreadsheets and manual binders as product lines and partners expand.
Document changes must translate into visible role expectations.
Audits, complaints, and supplier-facing actions need better structure.
The regulatory frameworks, guidance documents, and audit expectations that govern quality operations in this sector.
The FDA Current Good Manufacturing Practice regulation for dietary supplements — requiring written procedures, batch records, laboratory controls, and complaint handling for dietary ingredient manufacturers.
The FDA Food Safety Modernization Act CGMP and preventive controls rule — requiring hazard analysis, preventive controls, supply chain programs, and corrective action records for human food facilities.
The international food safety management standard and hazard analysis framework — requiring systematic identification of biological, chemical, and physical hazards with documented controls and verification records.
The Complere modules most commonly used by teams in this industry to manage compliance and quality operations.
Keep cGMP SOPs, batch production records, and product specification sheets version-controlled under 21 CFR Part 111 document control requirements — with electronic approval and automatic obsolescence handling.
Explore document controlDocument product complaints, batch failures, and HACCP deviations with structured investigation and corrective action workflows — meeting FSMA 21 CFR 117 preventive controls and supplier verification requirements.
Explore CAPA & deviationsSchedule and execute internal GMP audits and supplier qualification audits with structured checklists, finding tracking, and response workflows — supporting 21 CFR 111 and FSMA audit program requirements.
Explore audit managementMonitor CAPA closure rates, overdue batch record reviews, and training compliance in real time — giving quality managers continuous visibility into GMP operational health across the supplement production lifecycle.
Explore dashboards & KPIsExplore related modules, compliance topics, and guides to build a complete picture of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore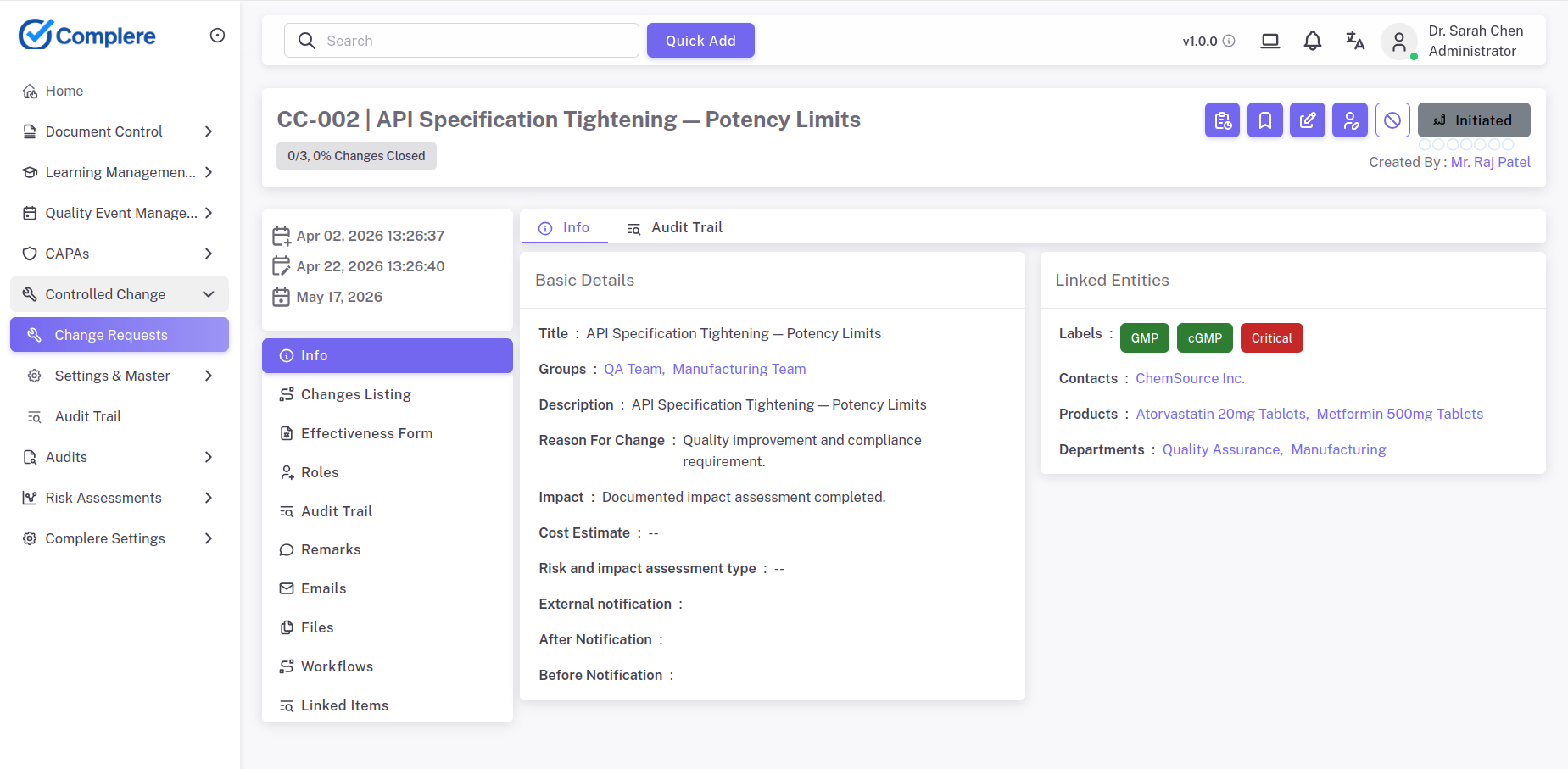
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreOur demos cover the GMP controls, supplier follow-up, change governance, and leadership visibility that matter most in this market.