CAPA & Deviations
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreThe regulated process for investigating quality issues, removing their causes, and preventing recurrence across a QMS.
CAPA is the backbone of quality governance. A well-run CAPA program shows inspectors that your organization does not just fix issues — it understands why they happened and prevents them from returning.

CAPA stands for Corrective Action and Preventive Action. It is the regulated quality process for responding to deviations, nonconformances, audit findings, customer complaints, and other quality issues in a structured, documented, and verifiable way.
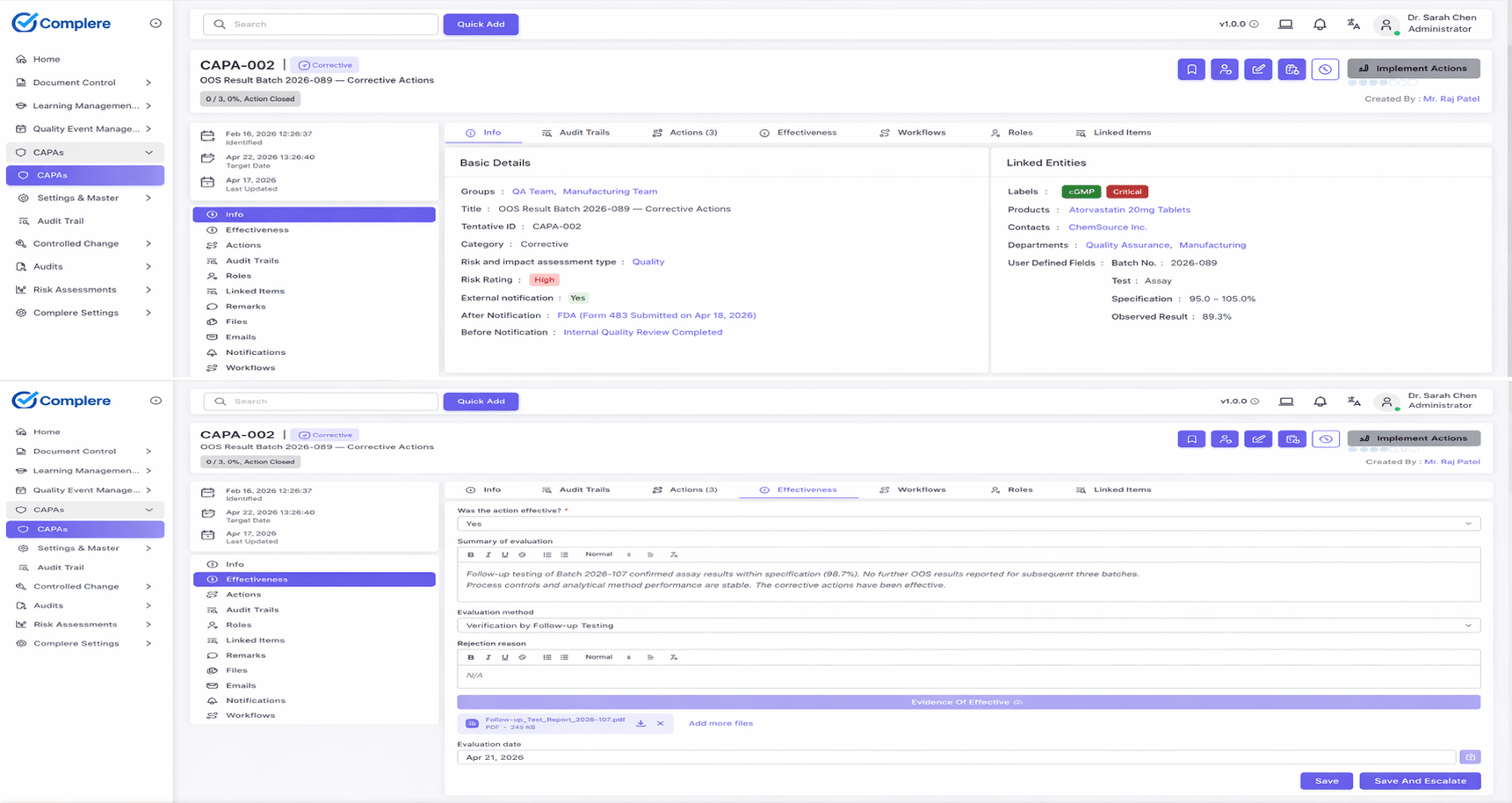
A CAPA record captures the problem, the investigation, the root cause, the actions taken to correct it (corrective action) and to prevent recurrence (preventive action), and an effectiveness check that confirms the fix worked.
CAPA is one of the most-inspected areas of any regulated quality system. FDA, EMA, MHRA, and notified bodies all look at CAPA records as primary evidence of a functioning quality culture.
A weak CAPA program — missing root causes, repeat deviations, late actions, missing effectiveness checks — is one of the top reasons regulatory bodies issue 483s, warning letters, and notified body findings.
Did you find the true root cause? Did your actions remove that cause? Did you verify the cause is actually gone?
CAPA requirements appear across most GxP regulations and standards:
A typical CAPA record moves through these stages:
Inspection-ready CAPA programs share common control patterns:
The Complere CAPA & Deviations module provides a controlled CAPA lifecycle with structured root cause capture, action plan tracking, and mandatory effectiveness checks.
Common questions about CAPA (Corrective and Preventive Action) sourced from regulatory references and inspection patterns.
An effective CAPA program captures meaningful signals, investigates root causes, documents proportional actions, and verifies effectiveness to prevent recurrence.
CAPA connects deviations to root cause analysis, documented action plans, verification activities, and cross-functional oversight rather than just recording events.
FDA 21 CFR 211, 21 CFR 820, EU GMP Chapter 1, and ICH Q10 expect documented CAPA governance with verification.
Inspectors review signal validity, root cause rationale, proportional actions, verification data, and whether the CAPA links to deviations, complaints, and management review.
Complere links CAPA findings to deviations, change control, document proofs, and audit trails, so inspectors see the full investigation-to-action story without manual compilations.
Explore related topics, modules, and compliance resources for a deeper understanding of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore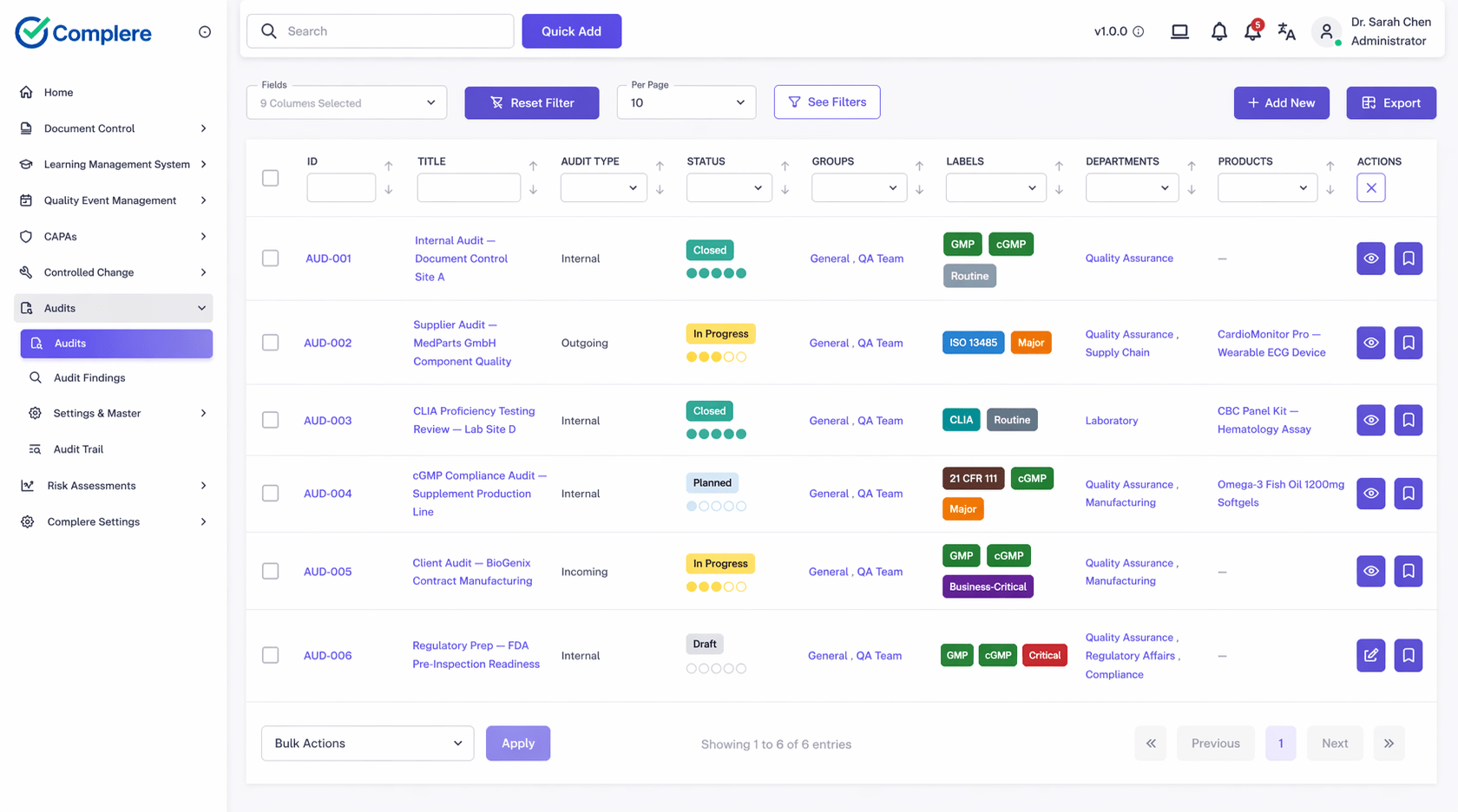
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreWalk through real CAPA investigations — root cause capture, action plans, effectiveness checks — connected to deviations, audits, and change control.