
Data Integrity & Audit Trails
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreA test result that falls outside registered specifications, method acceptance criteria, or established control limits — requiring formal investigation.
OOS investigation procedure matters because these records are among the most heavily scrutinized in pharmaceutical inspection. FDA's 2006 OOS guidance and the Barr decision shape how laboratories must respond when a result fails.

An OOS investigation in a GMP lab follows a defined two-phase procedure set by FDA's 2006 guidance and reinforced by the United States v. Barr Laboratories decision. The original failing result must be investigated before any retesting is considered, the procedure must be documented end-to-end, and batch disposition must be supported by evidence — not by repeated testing until a passing value appears.
At a high level, the procedure runs: lock the original result, execute Phase 1 (laboratory investigation), execute Phase 2 (full-scale investigation) if no assignable lab cause is identified, document a scientifically justified retest hypothesis when retesting is appropriate, and close with batch disposition and CAPA linkage for any systemic root cause.
See the In Practice tab for the full phase-by-phase steps, the Key Controls tab for the controls that keep the procedure inspection-ready, and the Regulatory Context tab for the cited FDA, EU GMP, USP, and MHRA references.
An Out of Specification (OOS) result is a test result that falls outside the specifications, acceptance criteria, or control limits established in a registered product specification, compendial monograph, or validated analytical method.
OOS is distinct from Out of Trend (OOT) — a result inside specification but trending in a concerning direction. Both require investigation, but OOS carries higher urgency and regulatory scrutiny.
The 1993 United States v. Barr Laboratories decision fundamentally shaped how pharmaceutical labs must handle failing results. The court ruled that retesting without investigation to dismiss an OOS is not permissible. FDA's 2006 guidance codified the expected two-phase investigation process.
OOS mismanagement — testing into compliance, inadequate investigation, late reporting — is one of FDA's most common data integrity findings. Warning letters frequently cite OOS handling as evidence of broader quality failure.
You cannot dismiss an OOS result simply by retesting until you get a passing value. The original result must be investigated to establish whether it was a lab error or a true product failure.
OOS handling expectations appear in:
FDA's 2006 OOS guidance defines a two-phase investigation process:
Robust OOS programs have:
OOS results are ingested as a dedicated Deviation sub-type in Complere, with structured phase 1 / phase 2 templates aligned to FDA 2006 guidance.
Common questions about Out of Specification (OOS) sourced from regulatory references and inspection patterns.
An OOS result is any analytical test result that falls outside the specifications, acceptance criteria, or limits established in approved methods or compendial standards. It requires a formal investigation to determine root cause and impact on product disposition.
QC initiates the laboratory investigation immediately upon confirming the OOS; if a lab error is not found, the investigation extends to manufacturing under QA oversight. Most pharma SOPs target closure within 30 days, with documented extension justification for complex investigations.
The 2006 OOS guidance requires a two-phase investigation: laboratory phase to rule out testing error, then full-scale Phase II investigation covering process, materials, and equipment. Retesting and resampling are restricted and must follow predefined scientific rationale.
OOS results fail specifications. OOT (out-of-trend) results fall within specifications but deviate from historical patterns and warrant evaluation. Atypical results show unusual analytical behavior even when within limits. All three require documented review under a robust quality system.
Complere connects OOS records to deviations, CAPA, batch records, and validated method history, so QA can reconstruct the full investigation timeline and disposition rationale without manual cross-referencing across spreadsheets and shared drives.
Explore related topics, modules, and compliance resources for a deeper understanding of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore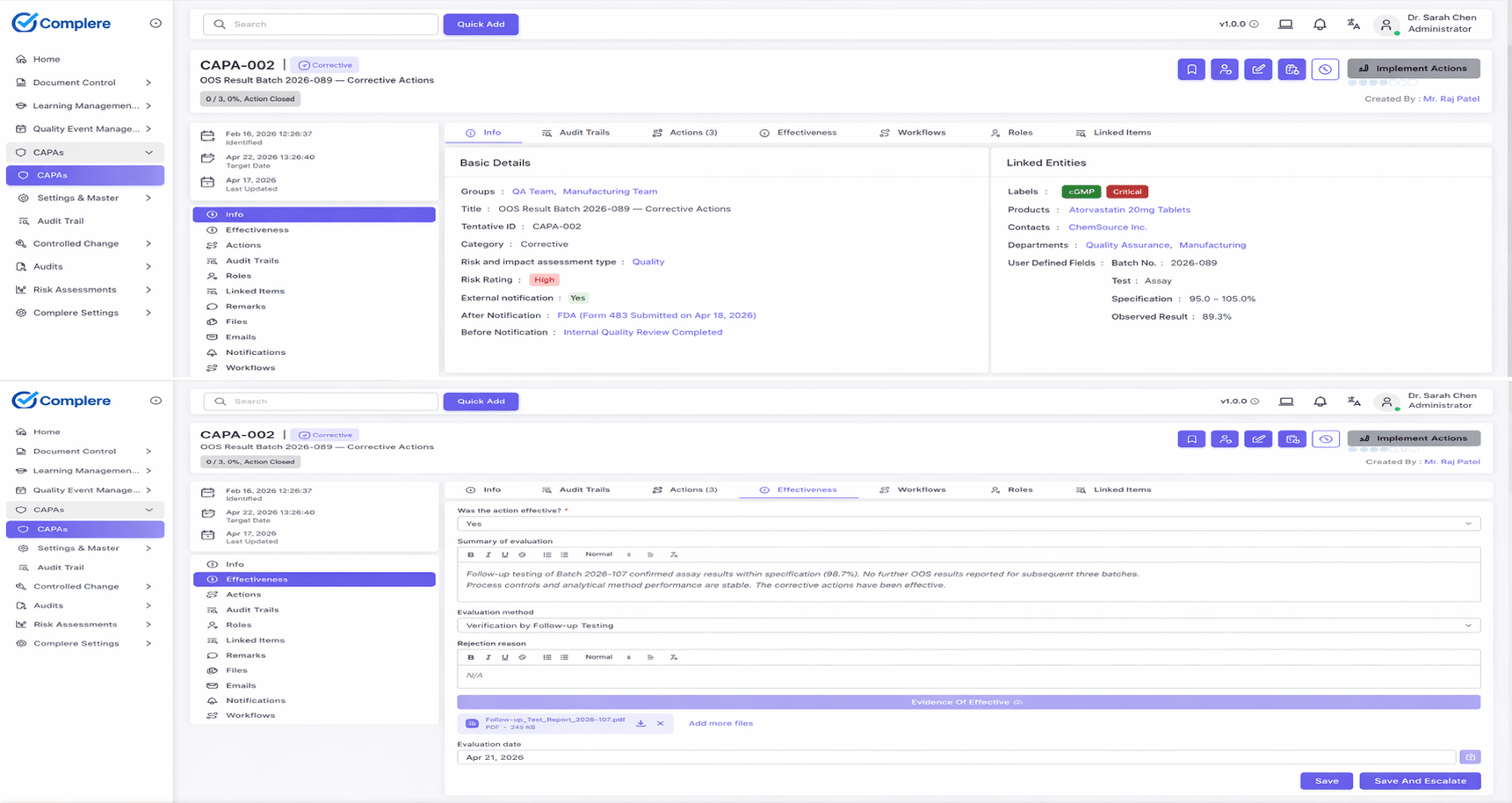
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreWalk through Complere's OOS-to-CAPA pipeline — structured phase 1 and phase 2 investigations with full audit trail and inspection-ready evidence.