CAPA & Deviations
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreAn unplanned departure from an approved procedure, specification, or standard operating practice — the starting point of a GMP investigation.
Every deviation is a signal. How an organization captures, investigates, and resolves deviations tells inspectors more about its quality culture than almost any other record type.

A deviation is an unplanned departure from an approved procedure, specification, master batch record, SOP, or other controlled document. It captures any event where the actual outcome differs from the documented, approved process.
Deviations are distinct from planned deviations (pre-approved changes with risk assessment) and from nonconformances (product-focused quality issues). Most GMP regulations use "deviation" for process and "nonconformance" for product, though terminology varies.
Deviations are the primary early signal of process drift, training gaps, equipment issues, or supplier problems. Inspectors read deviation trends to assess whether an organization sees its problems and manages them proactively.
Repeat deviations without corresponding CAPAs, long-open investigations, and superficial root causes are among the most common sources of 483 observations and warning letters.
Regulators expect deviations. What they scrutinize is how quickly and thoroughly you identified, escalated, investigated, and resolved them.
Deviation handling requirements appear in every major GMP framework:
Typical stages of a deviation record:
Strong deviation programs have:
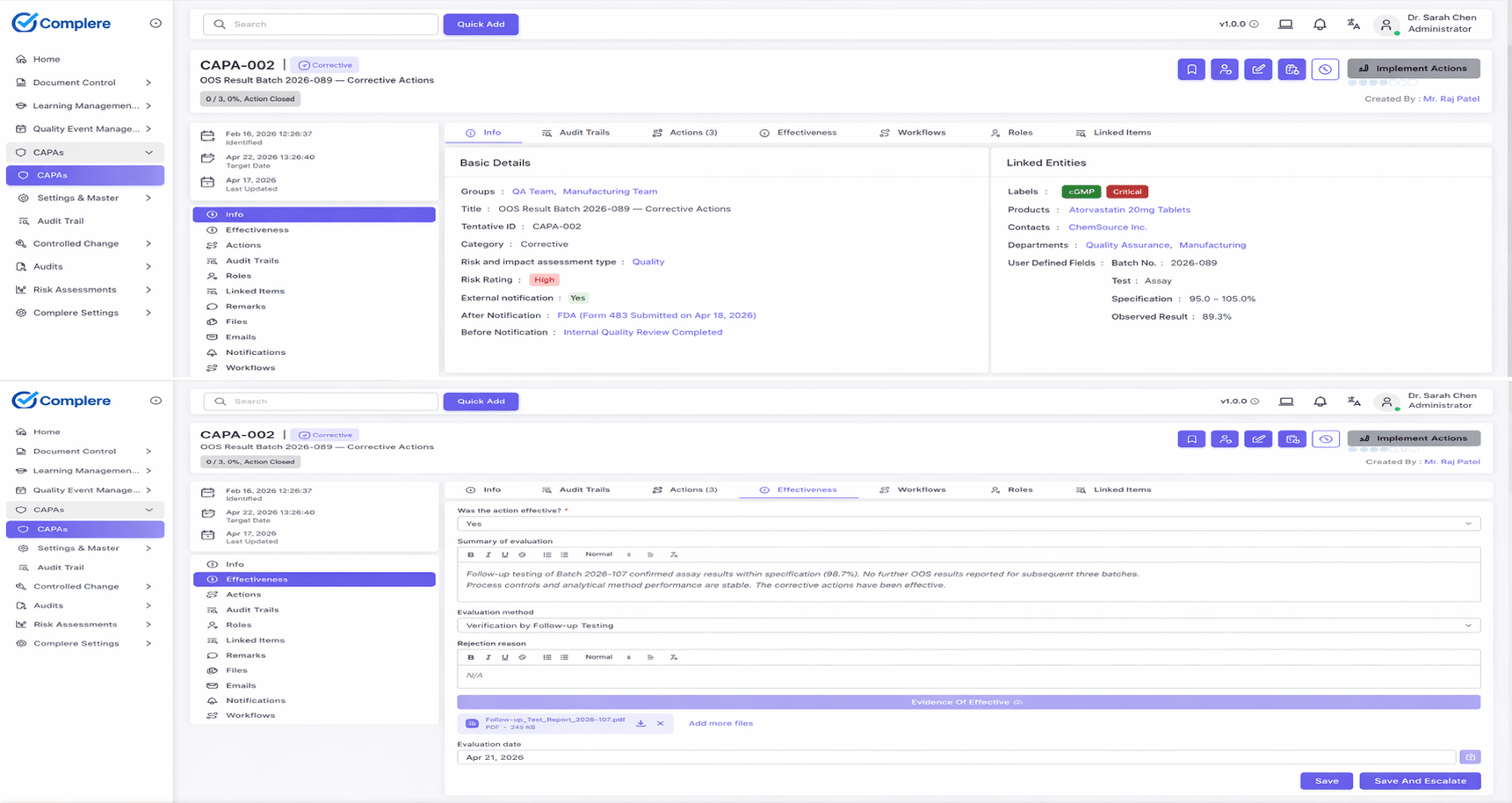
The Complere Deviation workflow provides intake through closure with structured investigation and direct CAPA linkage.
Common questions about Deviation sourced from regulatory references and inspection patterns.
Deviation management captures departures from approved procedures and triggers investigations, helping teams understand impact before product quality is compromised.
Deviation management probes GMP-impacting departures with classification, risk assessment, and documented follow-up, not just incident logging.
FDA cGMPs, EU GMP Chapter 1, PIC/S, and MHRA tie deviations to batch release, CAPA, and inspection readiness.
Inspectors examine timeliness, classification, root cause depth, linkage to CAPA, and the clarity of impact evidence.
Complere links deviations to supporting evidence, CAPA, change control, and training so you can show the entire investigation story in one traceable record.
Explore related topics, modules, and compliance resources for a deeper understanding of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreWalk through Complere's deviation intake, investigation, and CAPA linkage — with structured root cause and effectiveness evidence inspectors expect.