Hook
In a strong pharma quality system, a deviation should not live alone.
It should flow into investigation, Corrective and Preventive Action (CAPA), and change control without the record losing its history. When those steps are split across tools or handled manually, the team spends more time reconstructing the story than controlling the issue.
Real-world scenario
A batch issue is logged as a deviation. The investigation happens in one place, the Corrective and Preventive Action (CAPA) action plan is tracked somewhere else, and the related Standard Operating Procedure (SOP) update is handled later by email.
At first, each step looks complete. But when the QA lead asks how the event moved from detection to closure, the answer depends on memory, exports, and side conversations.
That is the real problem: the quality work happened, but the evidence chain did not stay together.
Why these records belong together
Deviation, Corrective and Preventive Action (CAPA), and change control are not separate compliance chores. They are three parts of the same quality response.
- A deviation captures the issue.
- Corrective and Preventive Action (CAPA) addresses the root cause and follow-up.
- Change control updates the process, document, or system so the issue is less likely to recur.
When those records are linked properly, the organization can explain not only what happened, but what it changed as a result.
What inspectors usually test
Inspectors rarely treat these records as isolated forms. They look for the chain.
| What they ask | What they are really testing |
|---|---|
| How did the deviation move to closure? | Whether the event, investigation, and follow-up are connected |
| Why was Corrective and Preventive Action (CAPA) opened? | Whether the root cause was credible and the action was justified |
| What changed after the Corrective and Preventive Action (CAPA)? | Whether the fix was implemented through controlled change |
| Was training updated? | Whether the affected people were brought into the change |
| Can you show the full history quickly? | Whether the system preserves the workflow, not just the final status |
Callout: the chain is the control
A deviation that never reaches Corrective and Preventive Action (CAPA), or a Corrective and Preventive Action (CAPA) that never reaches change control, is not a fully controlled quality response.
The risk is not only that something was missed. It is that the organization cannot prove the chain from issue to correction to implementation.
What fragmentation costs
When deviation, Corrective and Preventive Action (CAPA), and change control are split across tools, the cost shows up in more than one place.
- QA spends time stitching together evidence.
- Investigations take longer because context is scattered.
- Action ownership becomes harder to see.
- Document updates and retraining are easier to miss.
- Inspection response becomes a reconstruction exercise instead of a review.
That is why the issue is not just operational inconvenience. It is quality-system drag.
A short comparison
| Fragmented quality workflow | Connected quality workflow |
|---|---|
| Deviation, Corrective and Preventive Action (CAPA), and change records live in different places | The chain stays linked in one controlled workflow |
| QA rebuilds the story later | The story stays attached to the record |
| Document updates happen after the fact | Change control carries the update forward |
| Training can be missed | Training links to the change and stays visible |
| Inspection prep takes longer | Evidence is easier to retrieve and defend |
What a better system changes
A better system does not just store the separate records.
It keeps the relationship between them intact:
- the deviation stays attached to the investigation
- the Corrective and Preventive Action (CAPA) stays attached to the cause and the outcome
- the change stays attached to the reason it was raised
- the evidence stays visible from first event to final closure
That is what makes the workflow inspection-ready instead of just documented.
Download template
If your team wants to check whether these records are actually linked, a simple checklist helps.
The checklist should ask:
- Can we see the deviation, Corrective and Preventive Action (CAPA), and change in one chain?
- Is the reason for the change traceable back to the original issue?
- Do we know who owns each step?
- Is training or document update visible where it should be?
- Can QA show the full record path without rebuilding it manually?
That is a fast way to see whether the system is governing the process or just archiving it.
Complere fit
Complere is built to keep the quality chain intact, not scattered across files, emails, and spreadsheets.
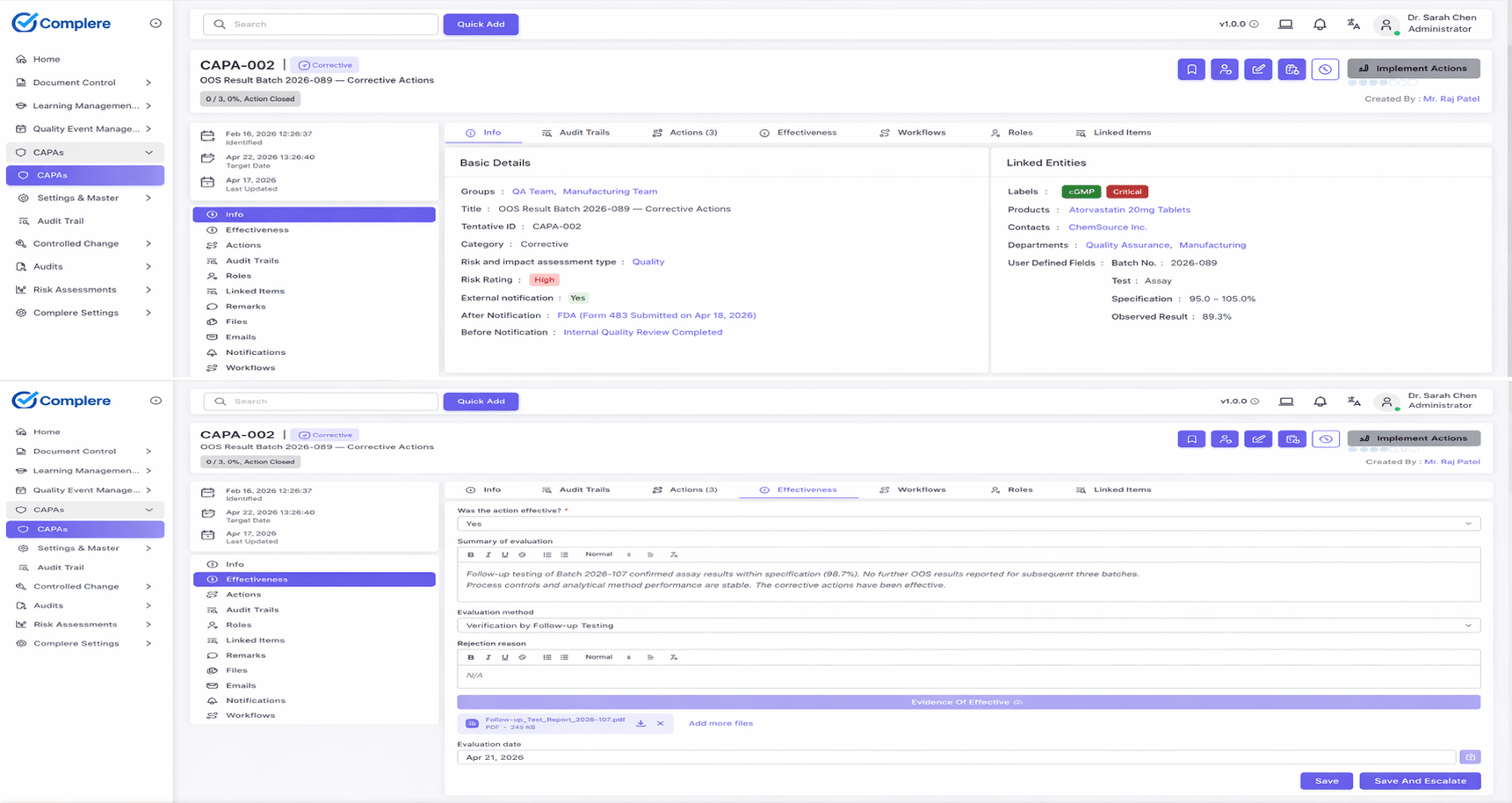
- The Events module supports deviations with investigation ownership, severity classification, root cause handling, attachments, electronic signatures, and Corrective and Preventive Action (CAPA) linkage.
- The Corrective and Preventive Action (CAPA) module carries action items, due dates, effectiveness review, and linked training evidence.
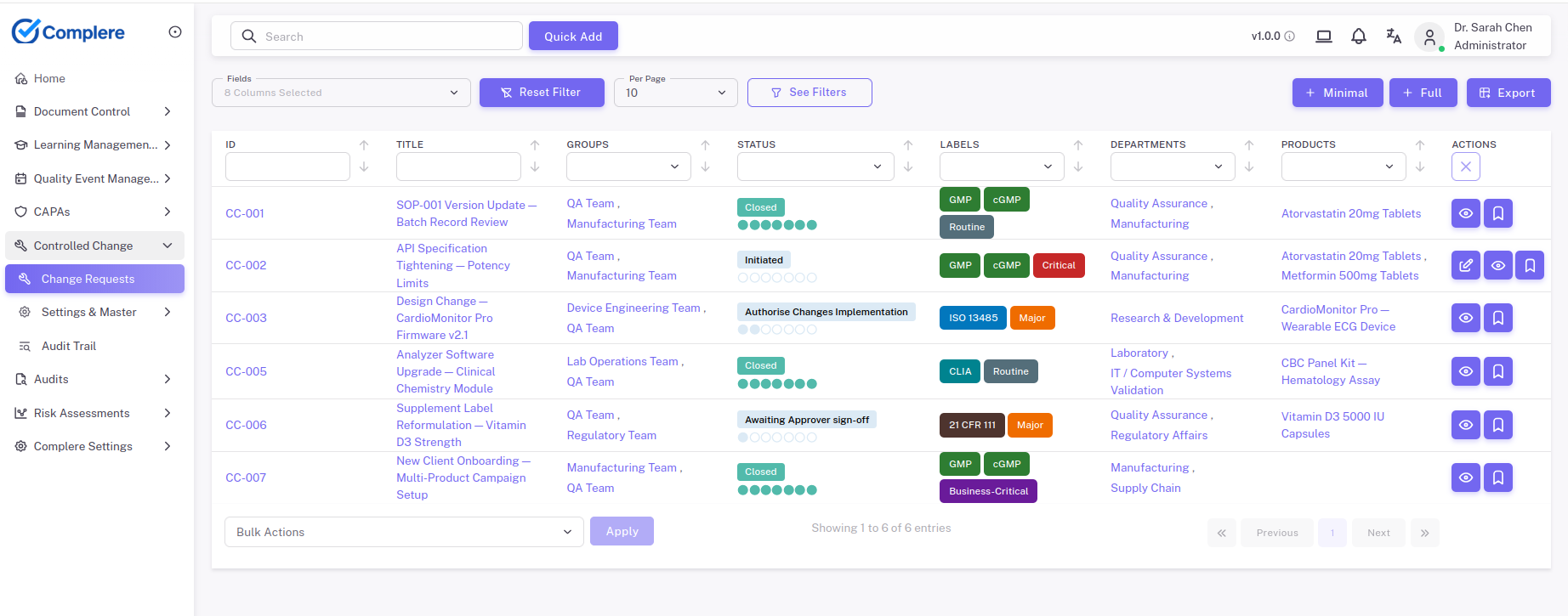
- The Controlled Change module supports impact assessment, document links, training links, approval, and post-change verification.
- Together, they keep the workflow history visible so quality teams can defend what happened without rebuilding the record trail later.
Complere does not just store the deviation, the Corrective and Preventive Action (CAPA), and the change request. It helps preserve the whole quality path as governed evidence so the team can explain what happened, what changed, and why it changed without rebuilding the chain later.
Closing thought
Deviation, Corrective and Preventive Action (CAPA), and change control work best when they behave like one quality response, not three separate records.
When they stay linked, the system is easier to manage, easier to inspect, and easier to trust.
Disclaimer
This article is a practical interpretation of GMP, ICH Q9, and Part 11-aligned quality expectations and is not legal advice. Teams should assess their own workflows, intended use, and validation approach.