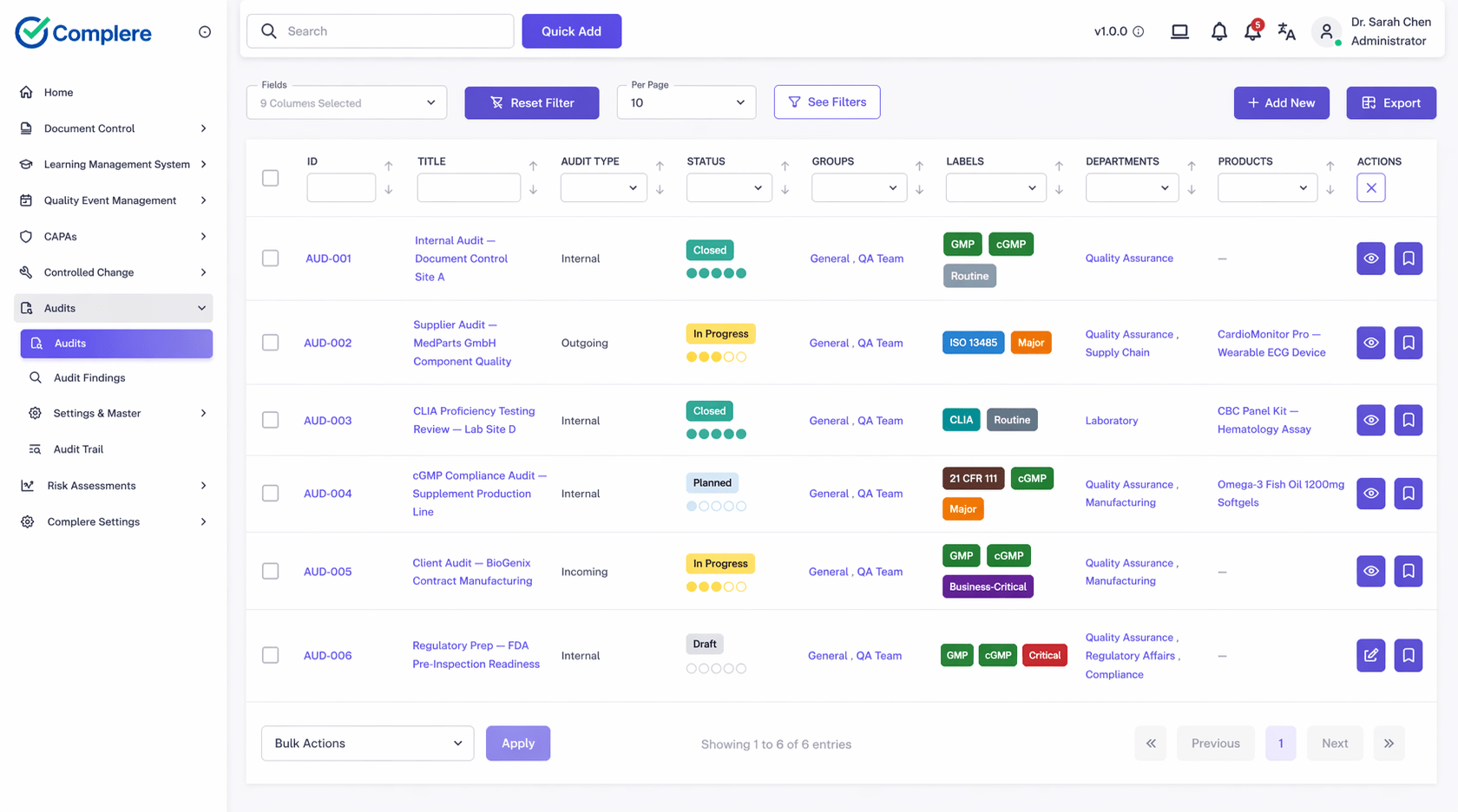
Audit Management Module
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreThe risk-based process for assessing, approving, monitoring, and re-qualifying suppliers of GxP materials, components, and services.
Suppliers are an extension of your quality system. Your inspection scope includes the materials they ship, the services they perform, and the controls they apply on your behalf.
Supplier qualification is the risk-based process for assessing, approving, monitoring, and re-qualifying suppliers that provide GxP materials, components, services, or outsourced activities. The objective is to establish a documented basis for concluding that the supplier can consistently deliver to specification under appropriate quality controls, and to maintain that basis over time.
Suppliers in a regulated context are treated as an extension of the firm's quality system. The regulator's view is that you can't outsource quality responsibility — you can outsource the activity, but you remain accountable for the outcome. Qualification, quality agreements, and ongoing oversight are the mechanisms that let you discharge that accountability.
The scope is wider than "manufacturing suppliers." It includes raw material vendors, API suppliers, contract manufacturers, contract laboratories, packaging suppliers, software-as-a-service vendors hosting GxP systems, calibration service providers, and contract sterilization providers — anyone whose work directly affects product quality or compliance.
When something goes wrong with a supplier, the regulator's first question is what you did to qualify them and what you do to monitor them. 'We trusted the supplier' is not an acceptable answer.
Several large recalls and regulatory actions over the past decade traced back to supplier quality failures: undeclared impurities in APIs sourced overseas, contamination in raw materials, contract manufacturers cutting corners on GMP, software vendors with inadequate validation discipline. Regulators tightened expectations in response.
Modern supplier qualification is no longer a one-time procurement step. It's a continuous oversight discipline that includes initial risk assessment, qualification audit, contractual quality terms, performance monitoring against defined signals, periodic re-qualification, and disqualification when signals warrant. Programs that still treat supplier qualification as an onboarding-only activity tend to surface in inspections through gaps the procurement team didn't realize were quality findings.
Inspector note: When I review supplier qualification at a firm, I trace one specific material from a recent batch back through the supplier chain. Was the supplier qualified at the time of receipt? Is the qualification current? Is the quality agreement in place? Have there been deviations or complaints involving the supplier, and did they trigger any oversight change? If those answers come together quickly with documented evidence, the program is working. If the team has to reconstruct the answers from emails and shared drives, it isn't.
Supplier qualification is anchored by both pharma and device frameworks plus contract-manufacturing and supply-chain provisions:
A complete supplier qualification program moves through recurring stages with controlled evidence at each:
Strong programs share consistent procedural discipline:
A supplier file with a 2018 questionnaire, a 2019 audit, and nothing since is a finding waiting to happen. Suppliers change ownership, lose certifications, get cited by other regulators, change processes. Without ongoing monitoring and periodic re-qualification, the file describes a supplier that no longer exists.
One thing to be upfront about: the dedicated supplier-management surface in Complere is still under construction. Today, your team runs supplier qualification through the modules that already carry the underlying quality work — audits, controlled documents, risk assessments, change control, events, and CAPA — because that's where the supplier activity actually lives. It's an honest cross-module approach, and the trade-off is that you'll think of supplier qualification as a discipline that spans the platform rather than a single supplier tab.
When your team audits a supplier, the audit runs as a structured supplier audit inside Complere. You plan the scope, schedule it, capture findings with severity, route signing through the right roles, and link any finding straight into a CAPA. Quality agreements, vendor questionnaires, and the approved-suppliers list itself live as controlled documents — templated, reviewed, electronically signed, version-controlled, and retained for the period your predicate rule requires.
Risk tier is treated as a real assessment, not a free-text label on a spreadsheet. You score critical suppliers against the methodology your quality system has agreed to, document the rationale, and let the tier drive the cadence and depth of everything that follows — audit frequency, contractual terms, the signals you watch for in performance monitoring. When something goes wrong, the path from a supplier-related deviation through investigation, root cause, and corrective action stays inside the same controlled workflow as the rest of your quality system.
When a supplier notifies you of a change under the quality agreement, that notification flows into change control with impact assessment, approvals, and any training-on-change your team needs to keep current. Across all of it, the supplier identity threads through audits, documents, risk assessments, change requests, events, and CAPAs, with the audit trail on every record so your team can reconstruct the full picture for an inspector — even though it doesn't yet live behind a single supplier dashboard.
What stays with your team: deciding the risk tier for each supplier, scoping the qualification work, running the audits and reviewing the agreements, and judging whether performance signals warrant restriction or disqualification. Complere supports the discipline with the same controlled workflows, role-based signing, and audit trail you use everywhere else; the supplier program around it stays yours, and we'll close the supplier-centric view as that module ships.
Common questions about Supplier Qualification sourced from regulatory references and inspection patterns.
Qualification is the documented basis for concluding that a supplier can consistently deliver to specification under appropriate quality controls — and the discipline for maintaining that basis over time. The depth of qualification scales to the risk the supplier poses: critical suppliers (APIs, sterilising agents, primary packaging) get the deepest treatment; lower-risk suppliers get proportionate treatment. The output is an entry on the approved-suppliers list with a defined scope of approval and a defined re-qualification cadence.
A supplier audit is one tool used within qualification — typically the most visible one, but not the only one. Qualification is the overall process: risk tiering, vendor questionnaires, regulatory and certification checks, historical performance review, sample testing, on-site audit (where warranted), quality agreement, and entry to the approved-suppliers list. Treating an audit as the qualification is a common gap; the audit alone doesn't qualify a supplier.
No. ICH Q10, EU GMP Chapter 7, and ISO 13485 §7.4 all expect a risk-based approach to audit depth and frequency. Critical suppliers — APIs, sterilising agents, contract manufacturers, contract laboratories supporting release testing — almost always warrant on-site audits at qualification and re-qualification. Lower-risk suppliers can be qualified through questionnaires, certifications (ISO 9001, ISO 13485, GMP certificates), historical performance, and sample testing. The trail from risk tier to audit depth needs to be visible on the record.
Not for every supplier, but strongly expected for critical and outsourced GxP activities. FDA's 2016 guidance Contract Manufacturing Arrangements for Drugs — Quality Agreements strongly recommends written agreements, and EU GMP Chapter 7 requires a written contract for outsourced activities defining the responsibilities of both parties. The agreement should cover scope of supply, change notification obligations, deviation and CAPA handling, complaints, audit rights, retention, and regulatory inspection notification.
Risk-based. Industry practice for critical suppliers is every two to three years for the full re-qualification cycle (including on-site audit), with continuous performance monitoring in between. Lower-risk suppliers can run on longer cycles or trigger-based re-qualification when a signal warrants. The 'approved at onboarding, forgotten ever since' pattern is a well-documented finding — suppliers change ownership, lose certifications, and modify processes; the qualification file needs to reflect the supplier that exists today.
Onboarding-only qualification with no ongoing oversight; quality agreement missing or expired; audit scope too narrow to support the qualification claim; approved-suppliers list not maintained or not consulted before purchasing; material received from a supplier whose qualification has lapsed or who has been restricted; supplier-related deviations that didn't trigger qualification review; and second-tier supply-chain visibility absent for critical materials.
Supplier identity (legal name, site, identifier); scope of approval (what materials or services the qualification covers); risk tier and the rationale; current qualification status (approved, conditional, restricted, disapproved); date of last qualification and date of next due re-qualification; reference to the active quality agreement; and links to recent audits, CAPAs, and significant supplier-related events. The list itself is a controlled document — single source of truth, retrieved before purchasing decisions, and kept current as supplier status changes.
Defined performance signals — material non-conformances, complaints, on-time delivery slippage, regulatory actions against the supplier, audit findings that recur — should trigger qualification review. The review's options are graduated: increased oversight (more frequent audits, tighter sampling), restriction to specific materials or sites, suspension pending corrective action, or full disqualification with the supplier removed from the approved list. Each option is a documented decision with rationale, signed by qualified personnel, and reflected in the approved-suppliers list before the next purchase.
Explore related topics, modules, and compliance resources for a deeper understanding of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore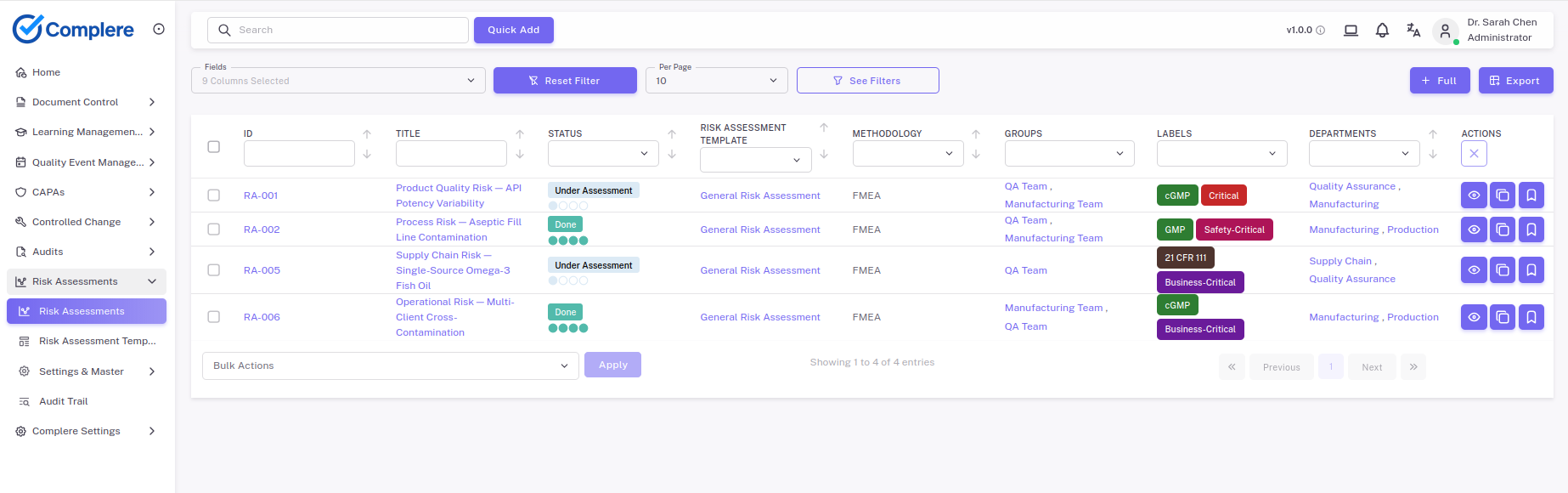
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreWalk through how Complere operationalizes this concept inside a validation-ready quality system.