Hook
Spreadsheets are comfortable because they are familiar. That is exactly why they stay in use long after a quality team has outgrown them.
In a regulated environment, comfort is not the same as control. Once records need traceability, review history, permissions, and defensible approvals, a spreadsheet becomes a weak place to manage the work.
Why the problem grows quietly
At a small scale, spreadsheets look efficient. One file, a few tabs, and everyone knows where to find things.
The problem starts when the work becomes shared:
- multiple versions start circulating
- comments and approvals live in different places
- updates happen without a clean audit trail
- no one can prove who changed what and why
That is usually when manual tracking stops being a convenience and becomes compliance debt.
Where spreadsheets break in practice
| Risk area | What happens in a spreadsheet | Why it matters |
|---|---|---|
| Version control | Files are copied, renamed, and emailed | The team loses a single source of truth |
| Audit trail | Edits are visible only in fragments | Reviewers cannot reconstruct the full history |
| Access control | Permissions are inconsistent | Sensitive data can be changed or overwritten |
| Review and approval | Sign-offs are detached from the record | The approval path is hard to defend |
| Retention | Old files are scattered across folders | Records are harder to retrieve during inspection |
What the audit path looks like
| Scenario | Spreadsheet-driven process | Controlled eQMS process |
|---|---|---|
| Opening a deviation | QA searches through one file, then another | The record opens with linked context attached |
| Reviewing the change | Version history is scattered or incomplete | Changes, approvals, and rationale sit in one workflow |
| Checking CAPA follow-up | Reminders depend on people and email | Actions are tracked and escalated inside the system |
| Showing evidence | The team assembles proof after the fact | The evidence already lives with the record |
Why auditors care
Auditors are not looking for a tool preference. They are looking for evidence that the process is controlled.
If a quality record depends on a spreadsheet, the burden shifts to the company to prove that the file is protected, the changes are traceable, and the final record is trustworthy. That is a higher bar than most teams realize.
The issue is rarely the spreadsheet itself. The failure happens when teams can no longer reconstruct the record quickly, show version history clearly, or explain why a specific change was made.
Regulatory basis
The concern is not theoretical. FDA expectations around electronic records, signatures, traceability, and data integrity make uncontrolled manual files a weak foundation for regulated work.
In practice, inspectors look for:
- a clear record of who did what and when
- controlled changes and approvals
- retrievable and legible records
- evidence that the final record is trustworthy
That is why spreadsheet-based control becomes hard to defend once the process is shared across people, sites, or products.
For this article, the point is not that every spreadsheet is non-compliant. The point is that once a spreadsheet becomes the operational system of record, it has to carry compliance weight it was never built to carry.
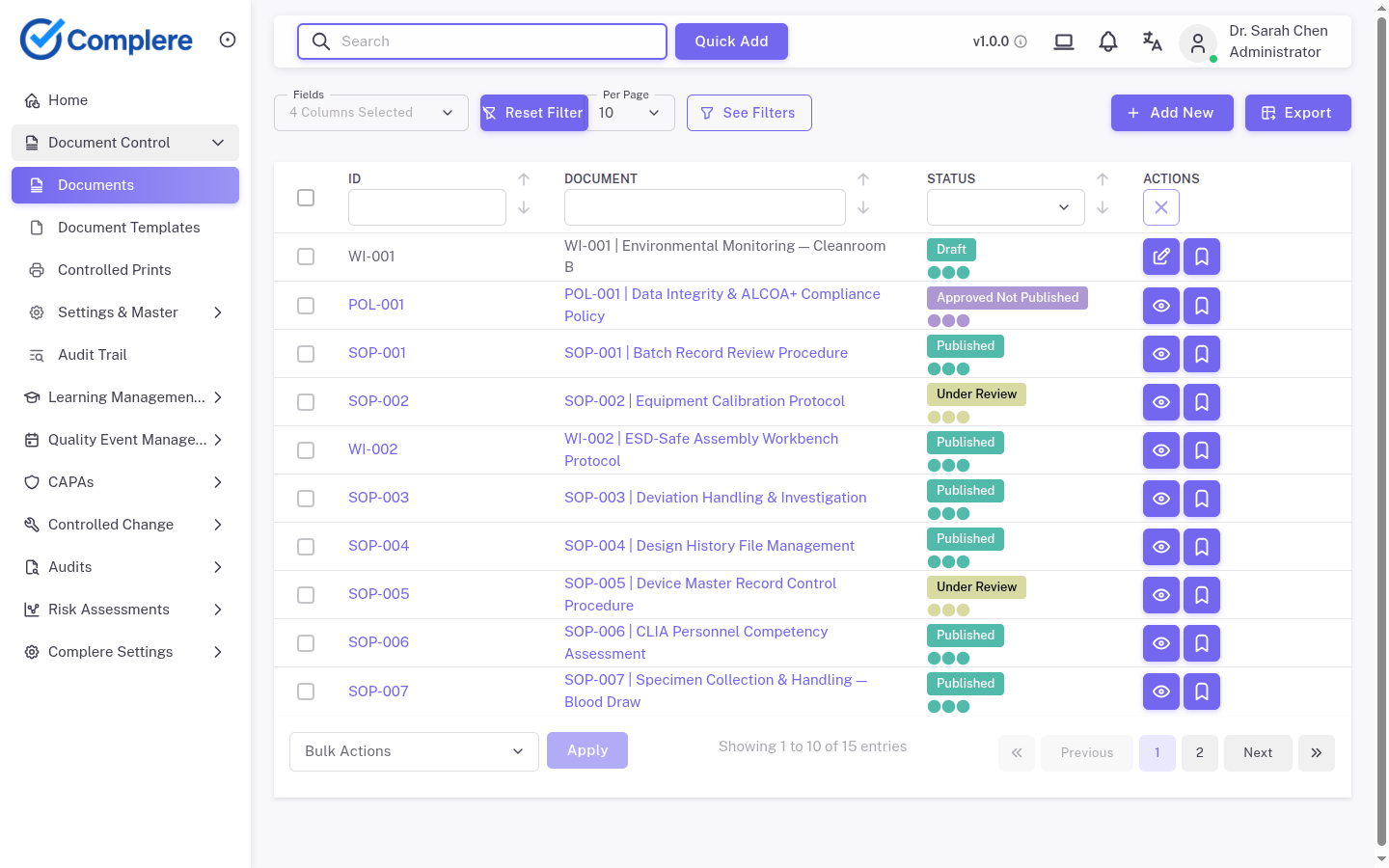
What a better system changes
A controlled eQMS does not just replace the file. It changes the way the work is governed.
The record lives in one place. The history stays attached to the action. Approvals sit inside the workflow. Review and follow-up are visible.
The goal is not to make the spreadsheet prettier. The goal is to move the work into a controlled path where evidence is created as the work happens.
That is the difference between a file that stores information and a system that supports compliance.
Download template
If your team still manages quality work in spreadsheets, the next practical step is to make the risk visible. A short checklist helps teams see where the process depends on manual files, manual reminders, or file-based approvals.
The spreadsheet risk checklist can be used as an internal review aid before a team starts comparing systems or planning a migration.
Complere fit
For teams still relying on spreadsheets, the real issue is not the spreadsheet itself. It is the lack of governed workflow behind it.
Complere helps move that work into controlled modules so quality teams can stop stitching records together after the fact.
Disclaimer
The cost and process examples in this article are illustrative. They are meant to show how manual quality work creates hidden effort and risk, not to replace site-specific validation or compliance review.
Closing thought
If the team spends more time explaining the spreadsheet than using it, the process has already outgrown the tool.
The question is not whether spreadsheets are useful. The question is whether they are still defensible in a regulated quality system.