Hook
Hybrid does not automatically mean broken.
It does mean the story can live in two places at once, and that is usually where pharma teams start to lose the thread.
Real-world scenario
It starts in a way that feels practical.
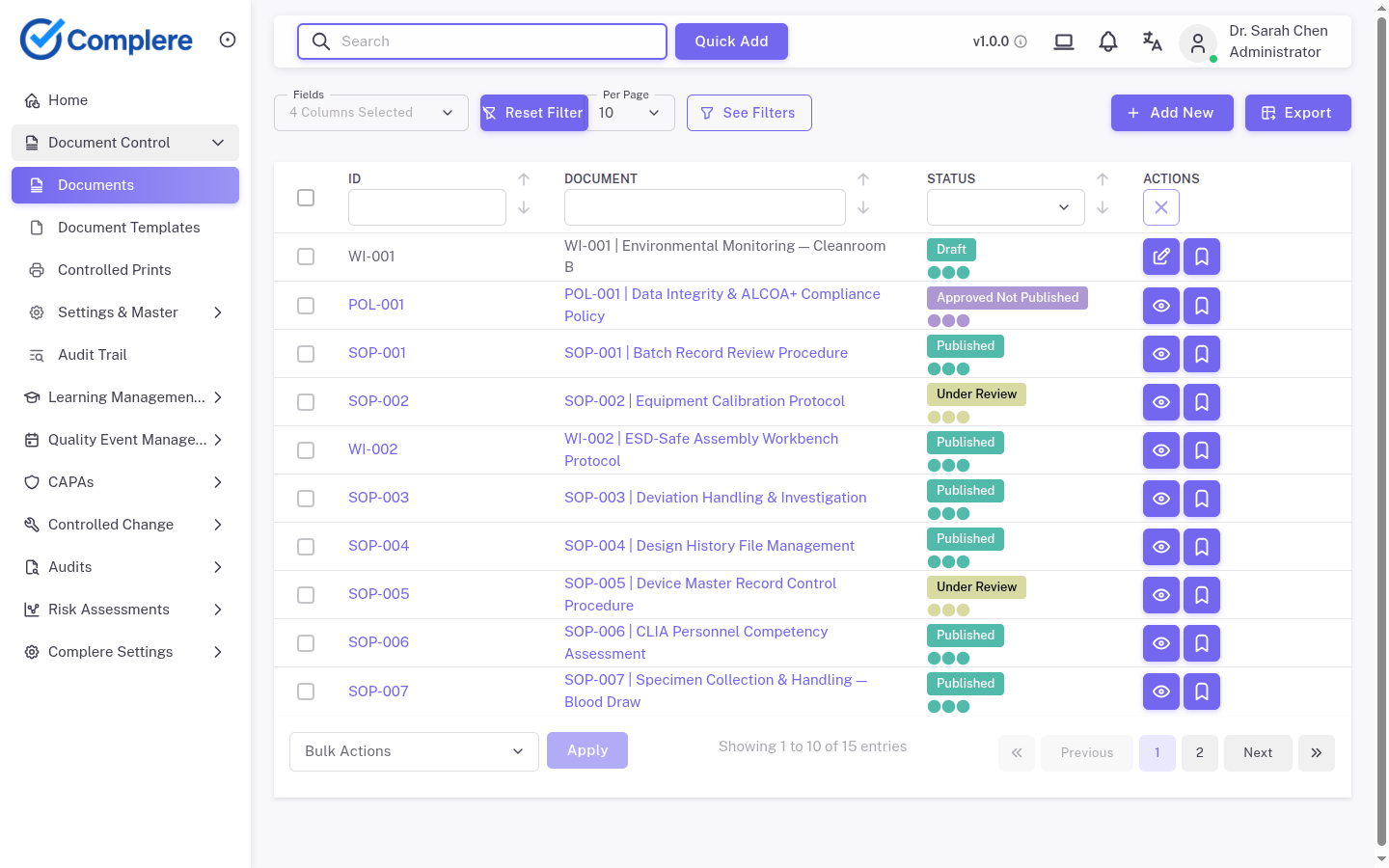
A procedure lives in a binder on the floor.
Training is tracked in a spreadsheet or Learning Management System (LMS).
The deviation was logged by email.
Someone approved the change from a shared drive.
The paper copies still sit in a drawer because “that is what people are used to.”
Most days, nobody complains.
Then an inspector asks for one clean path:
- what changed
- who approved it
- who got trained
- which version was in use
- what happened after the deviation
That is when the hybrid setup stops looking tidy.
What actually fails in a hybrid system
| Area | What happens in a hybrid setup | Why it fails under inspection |
|---|---|---|
| Document control | One version is signed on paper, another sits in a shared drive, and someone still keeps a local copy “just in case” | The team wastes time proving which version was current and who actually saw it |
| Training records | Completion is logged in one place while the updated procedure lives somewhere else | The signature exists, but the link to the actual change is weak |
| Deviation follow-up | The event starts in an email thread or spreadsheet and ends in a different file | The sequence is hard to reconstruct when someone asks what happened next |
| Audit trail | Some steps are electronic, some are handwritten, and some only live in memory | The story is fragmented, so the record feels incomplete even when the pieces exist |
| Retrieval | You need exports, screenshots, and a few explanations from different people | Inspection time gets spent rebuilding context instead of showing the record |
Callout: the problem is split evidence
Hybrid is not the sin. Split evidence is.
If the approval, the change, the training, and the current version do not point to each other, the record may exist, but it does not travel well in an inspection.
Why inspections expose it
FDA’s Part 11 guidance does allow paper and electronic components to coexist in a hybrid situation, but that is only helpful if the predicate-rule requirements are still met and the content and meaning of the record are preserved.
That is the part teams miss.
The real inspection test is not whether you have some paper and some software. It is whether the system still lets you produce a trustworthy, human-readable story without scrambling.
MHRA’s data integrity guidance points in the same direction: records have to stay reliable across the whole lifecycle, not just look fine when they are filed away.
What a connected system does differently
In a connected setup, the team does not have to chase the story.
- the current version is obvious
- the approval history is tied to the record
- the training stays linked to the reason it was needed
- the change remains visible after the fact
- inspection response stops depending on memory and exports
That is the practical difference. Not “paper bad, digital good.” Just less splitting, less guessing, less reconstruction.
What Complere changes
This is where Complere helps, without pretending to be magic.
Complere keeps the record, the approval, and the follow-up tied together so the chain does not break when the work moves.
- When a document changes, the update path stays visible.
- When training is needed, it stays attached to the change that caused it.
- When an inspection asks for the story, the team is not starting from a pile of exports and screenshots.
We did not build it to replace one pile of paper with another digital pile. We built it so the record still makes sense when someone asks hard questions about it.
Download template
If your site still runs hybrid, use a short inspection checklist and be blunt with the answers.
Ask:
- Can we show the current approved version without a scavenger hunt?
- Can we prove who was trained on it and when?
- Can we trace the deviation or change back to the current record?
- Can we explain the handoff between paper and digital without guessing?
- Would this story hold up if an inspector asked for it right now?
If the answer is fuzzy, the hybrid setup is already showing its seams.
Closing thought
Hybrid systems usually work right up until someone needs the whole story at once.
That is why inspections are uncomfortable. They do not test whether the pieces exist. They test whether the pieces still belong to the same record.
Disclaimer
This article is a practical interpretation of regulated recordkeeping and inspection expectations and is not legal advice. Teams should assess their own workflows, intended use, and validation approach before relying on a hybrid system.