Hook
A CDMO is not buying an eQMS just for itself.
It is buying for the sponsor, the auditor, and the next handoff that nobody wants to explain twice.
Real-world scenario
A sponsor sends a change request.
The site needs to update a procedure, retrain the team, and keep the quality agreement aligned.
That sounds straightforward - until the evidence starts living in different places.
The quality agreement is in email.
The change request is in one system.
Training is in another.
The sponsor wants a clean explanation, but the team is already piecing the story together from folders and screenshots.
That is where a lot of CDMO setups start to feel heavier than they should.
And you usually do not get much time to assemble that answer. You are expected to show the path across both the site and the sponsor view.
What CDMOs should demand
If you run a CDMO, a connected eQMS should help you do a few things without turning every request into a scavenger hunt:
- show the current quality agreement or controlled commitment quickly
- keep sponsor-facing change requests tied to the real implementation path
- make external quality work visible without giving away control
- keep training and approvals linked to the change that caused them
- preserve the audit story across site, sponsor, and third-party records
What actually breaks in a weak setup
| Area | Weak setup | CDMO-ready setup |
|---|---|---|
| Quality agreements | Stored as PDFs in email or shared drives | Controlled, current, and easy to retrieve |
| Sponsor requests | Handled in inboxes or spreadsheets | Assigned, tracked, and visible in the workflow |
| External access | Guests get attachments and status updates by email | External parties can see the right record without controlling it |
| Change control | Site-only record with loose follow-up | The sponsor impact is visible and the follow-through stays attached |
| Inspection support | Team rebuilds the story from different sources | The evidence chain is already connected |
Callout: external controls fail when everyone keeps their own story
If the sponsor, the site, and the quality team all store a different version of the same change, the record is already split.
That is the part inspectors and auditors notice fast.
Why this matters more for CDMOs
CDMOs live on handoffs.
They move product, records, actions, and decisions across more than one team. When that happens, the system has to be good at keeping context intact.
FDA’s quality-agreement guidance makes that responsibility explicit: the parties involved in contract manufacturing need clear, documented responsibilities so the work does not get fuzzy at the edges.
That is why an eQMS for a CDMO is not just about internal order. It is about being able to show the sponsor what changed, who approved it, and how the follow-up stayed controlled.
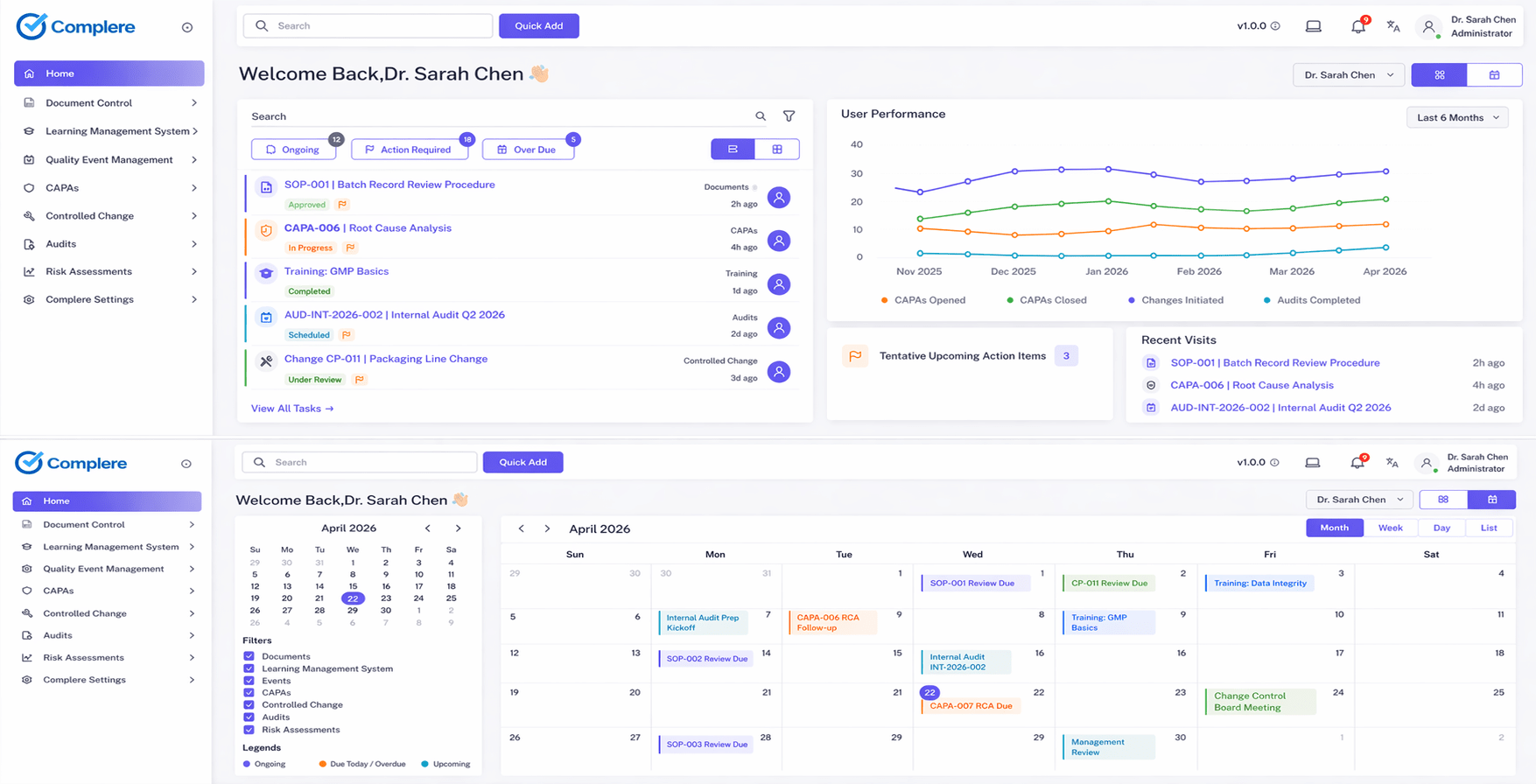
What Complere changes
Complere helps by keeping the sponsor-facing quality story connected instead of scattered across email, shared folders, and side trackers.
- external parties can be brought into the process without turning them into system owners
- sponsor requests and follow-up stay attached to the quality record
- quality agreements and related documents are easier to keep in one controlled path
- training, change, and approval history stay visible when the site has to explain the chain
We did not build this to add another inbox. We built it so the CDMO can keep one defensible story across the sponsor relationship and the site workflow.
Download template
If you are reviewing an eQMS for a CDMO, use a short checklist and be strict with it.
Ask:
- Can the sponsor-facing record be shown without a long email trail?
- Can the team prove who approved the change and when?
- Do training and change records stay linked?
- Can external parties see what they need without taking over the system?
- Would the audit story still hold if a sponsor asked for it today?
If the answer is shaky, the system is not ready for external work.
Closing thought
CDMOs do not win trust by having more documents.
They win trust by making the handoff clean enough that nobody has to rebuild the history later.
In a CDMO setup, control is not just internal. It is shared. And if the system cannot hold that, the record splits.
Disclaimer
This article is a practical interpretation of regulated supplier and contract-manufacturing expectations and is not legal advice. Teams should assess their own quality agreements, intended use, and workflow design before changing their system.