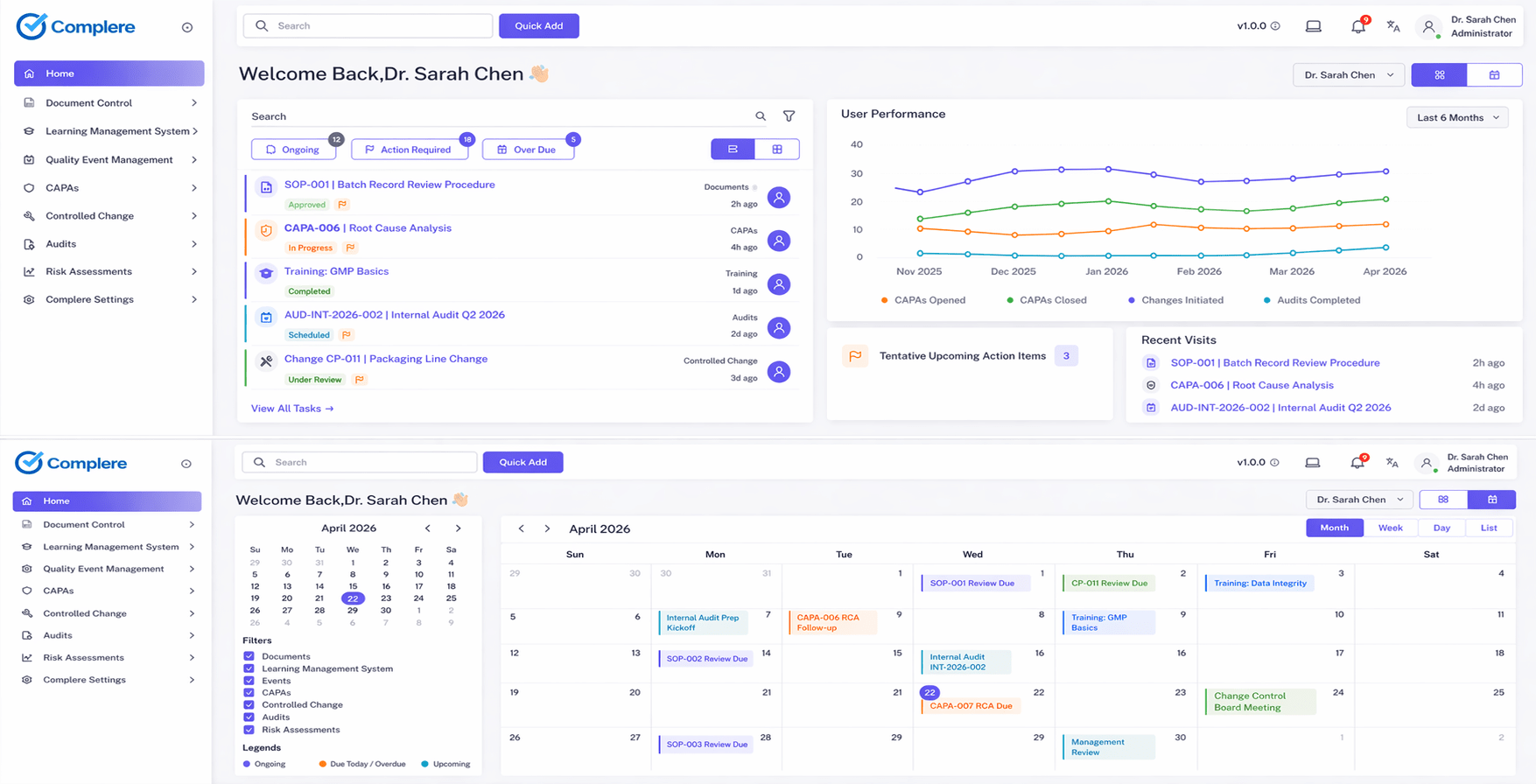
Hook
Most pharma teams do not go looking for an eQMS because they love software.
They go looking because the current setup starts to fall apart when you need the record to hold together under pressure.
Real-world scenario
It usually starts looking harmless.
Paper folders. Shared drives. A few approval emails. Maybe one extra tracker "just to be safe."
Then production gets busier and the mess stops being subtle.
An SOP gets updated, but operators are still pulling old printed copies from a drawer. Training records look green on paper, but the floor team is already running batches to a newer procedure. A deviation needs CAPA, and now someone is trying to remember which tracker actually owns the follow-up. Change control becomes a round of reminders instead of one clean path.
That is usually the point where the question changes.
Not "does the QMS exist?"
More like: can we defend this without dragging three people into a room and rebuilding the story from memory?
What actually changes
| Area | Traditional QMS | eQMS |
|---|---|---|
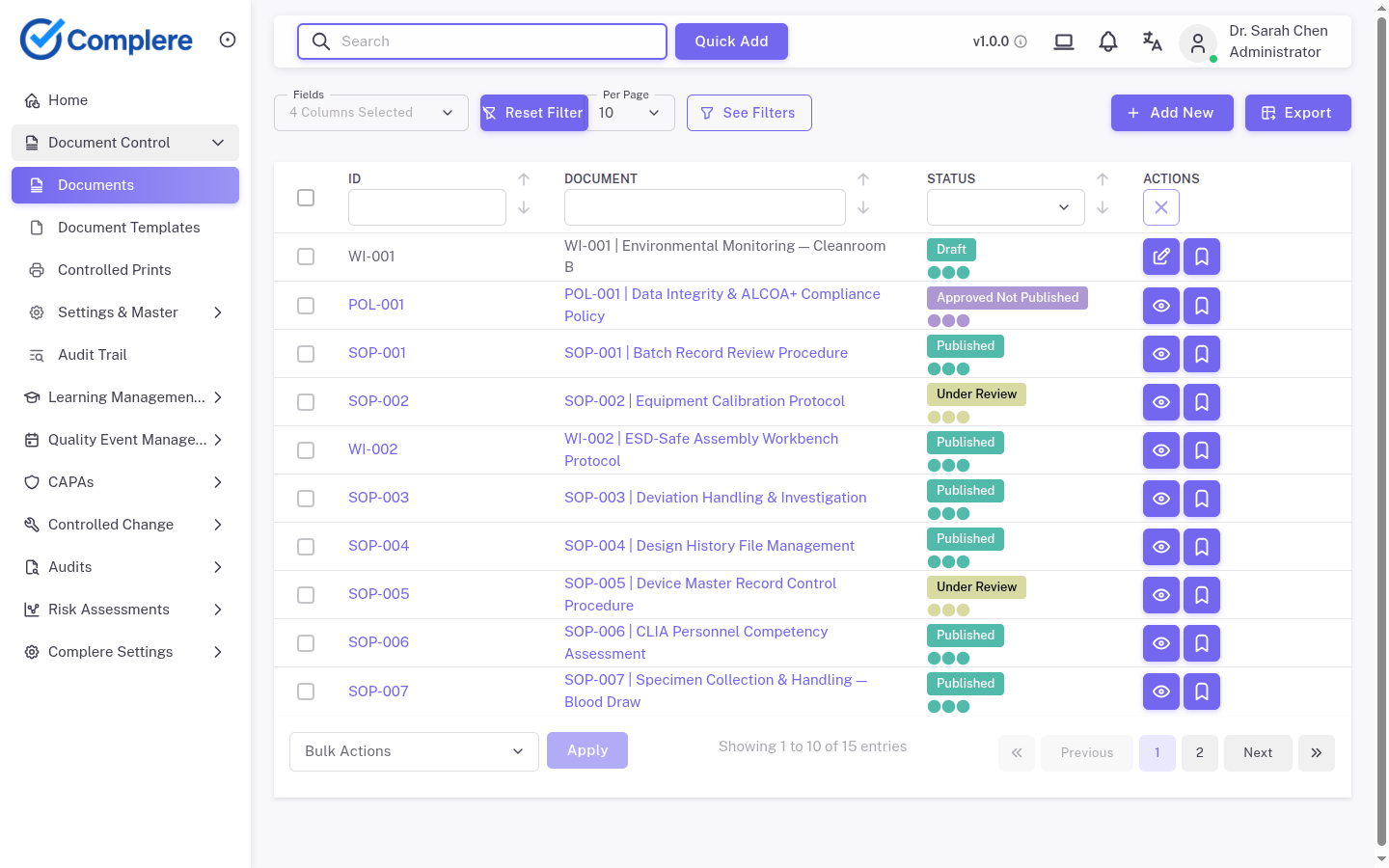
| Document control | Someone is still hunting for the latest signed copy, and the version history is scattered across folders and inboxes | Controlled versions, workflow approvals, and traceable history |
| Audit trail | Logs exist, but the team still has to reconstruct the sequence after the fact | Activity stays attached to the record and is easier to review |
| CAPA | Separate trackers, reminder emails, and overdue actions QA has to chase | Linked corrective action with ownership and effectiveness review |
| Change control | Email chains, local edits, and side conversations hide the real implementation path | Structured impact assessment and implementation verification |
| Training | Completion sheets can drift away from the actual procedure in use | Evidence tied to the actual document or process change |
| Inspection support | A last-minute fire drill to collect exports, screenshots, and explanations | Retrieve the story directly from the workflow |
Callout: what the first audit question sounds like
Can you show the path from deviation to CAPA to change without opening four systems?
If the answer is export files and explanation, the system has already leaked control.
Callout: the real shift
The big change is not that records become electronic.
The big change is that the quality process becomes connected enough for the team to defend it without pulling evidence from four different places.
If the system still needs manual reconstruction, the digitization is cosmetic.
Decision risk: what a poor eQMS choice leaves behind
A weak eQMS selection does not solve the old problem. It usually repackages it.
That shows up as:
- the same audit findings in a digital wrapper
- more QA time spent chasing status instead of controlling quality
- duplicate trackers kept "just in case"
- training, CAPA, and change still living in separate places
- revalidation work when the team has to switch again later
If the software does not change the workflow, it only changes the screen.
What changes for QA and compliance
For QA, the eQMS changes the daily burden:
- fewer handoffs to chase
- less manual evidence stitching
- clearer ownership of quality actions
- faster review of active and overdue items
- better visibility when something drifts
For compliance, the eQMS changes what can be defended:
- approvals are traceable
- record history is easier to show
- changes are tied to actions
- training stays aligned with the current state
- audit response becomes faster and less fragile
What a bad migration looks like
Moving from QMS to eQMS does not automatically improve control.
A bad migration gives you:
- old process habits inside new software
- unused modules
- duplicate trackers kept "just in case"
- team confusion over where the real record lives
- audit prep that still depends on export files and memory
That is why the software decision matters less than the workflow design.
What Complere changes
You are not buying modules. You are deciding whether the quality story stays intact when the work moves.
Most QMS platforms turn into a bundle of modules. Complere is built to keep the quality process from splitting apart as the work moves.
That matters because once deviation, CAPA, and retraining start living in different places, teams end up chasing their own history.
- Complere keeps the issue, investigation, action, and verification in one governed path.
- Complere keeps deviation, CAPA, change, and training from drifting apart as the record moves.
- Complere flags the SOPs and training evidence that should move with a change instead of leaving that to memory.
- Complere keeps QA inside the live workflow instead of pushing them into export-and-explain mode.
- Complere gives pharma teams a way to defend the process without rebuilding the evidence chain from exports and memory.
That is the point of the platform. It is not just digitizing quality forms. It is keeping the workflow intact so QA is controlling the work, not reconstructing it later.
If the record still needs to be stitched back together by hand, the software has not really changed the job.
Download template
If your team is deciding whether a QMS is enough or whether an eQMS is necessary, use a simple comparison sheet.
The sheet should ask:
- Can the current system preserve the full evidence chain?
- Are approvals, changes, and training actually linked?
- How much manual work does QA still do to defend the record?
- Can the team show audit history quickly?
- Does the system reduce reconstruction work or just move it into software?
That is the cleanest way to see whether you need a digitized QMS or a real eQMS.
Closing thought
QMS to eQMS is not just a technology upgrade.
It is a move from managing records to governing the quality process itself.
If the system cannot keep the chain intact, it has not really changed the work.
Disclaimer
This article is a practical interpretation of regulated quality-system requirements and is not legal advice. Teams should assess their own workflows, validation approach, and intended use before making a system change.