Hook
ALCOA+ is one of those phrases people repeat often and explain poorly.
In practice, it is not a slogan. It is a test of whether a record can be trusted when someone needs to review it later, defend it during an inspection, or use it to support a quality decision.
Real-world scenario
A quality team may collect information carefully, but still lose trust in the record if data is split across spreadsheets, emails, and local folders.
The issue is usually not one dramatic failure. It is a slow drift: an update lands in the wrong version, a comment is removed from context, or the record no longer tells a complete story.
That is where ALCOA+ stops being theory and starts becoming a daily operating standard.
What ALCOA+ means
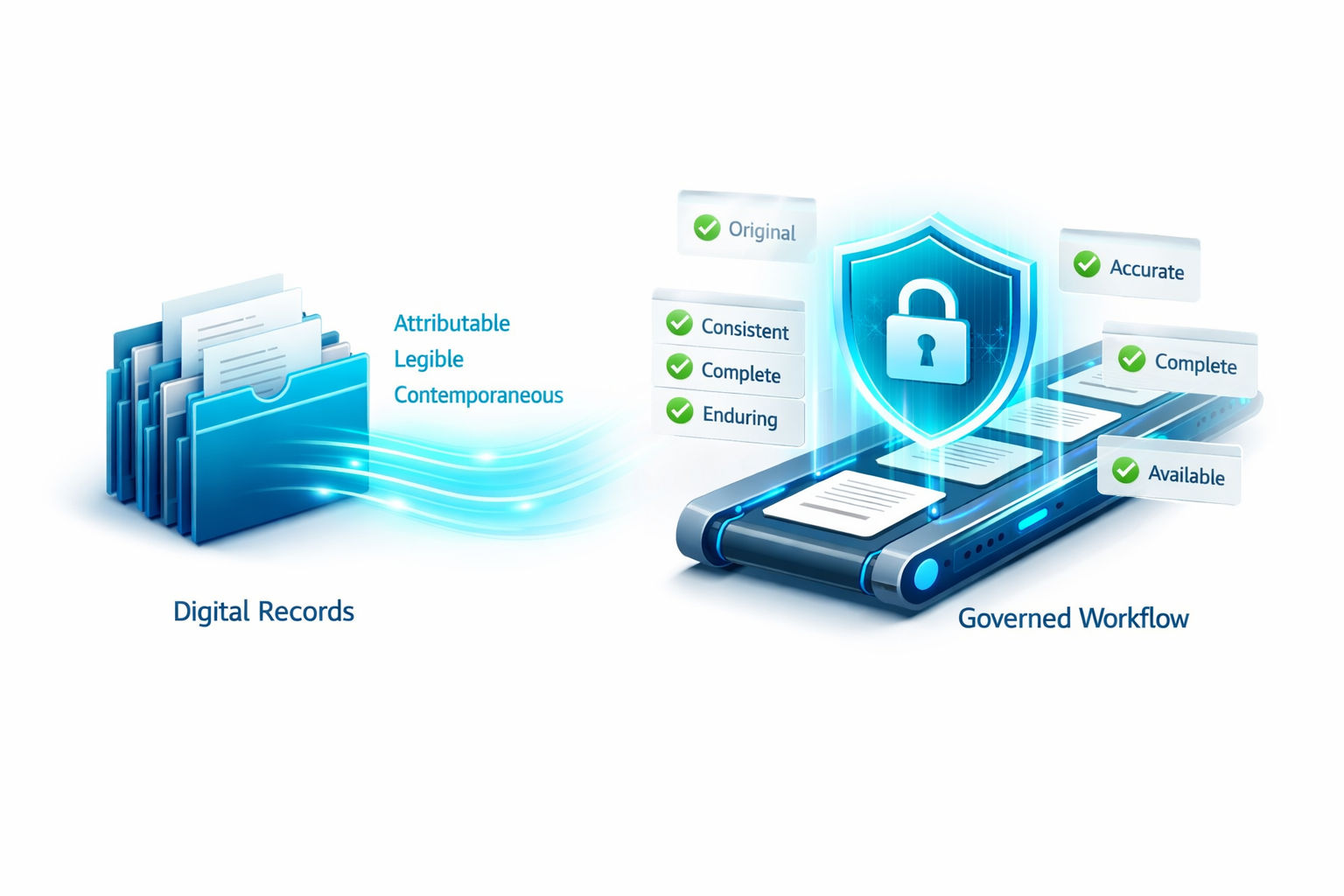
ALCOA+ is the data integrity framework used to judge whether records are reliable enough for regulated work.
The letters stand for:
- Attributable: you can tell who created or changed the record
- Legible: the record can be read and understood
- Contemporaneous: the record was created when the work happened
- Original: the source record is preserved, or a true copy is available
- Accurate: the content is correct
- Complete: nothing material is missing
- Consistent: the record follows the expected sequence and logic
- Enduring: the record remains usable over time
- Available: the record can be retrieved when needed
Recent FDA materials continue to frame data integrity around the same core idea: data must be reliable, complete, and maintained in context. That is not a narrow electronic-records problem. It applies to paper, digital, and hybrid environments.
Why it matters
FDA data integrity guidance and related quality materials keep returning to the same message: records have to support trustworthy decision-making.
That matters because the record is not just evidence for an auditor. It is the basis for quality actions, product decisions, and follow-up work.
If the record is weak, the decision becomes weak with it.
What teams usually get wrong
Most data-integrity problems are not caused by people trying to cheat the system. They are caused by workflows that make records easy to fragment.
- Common assumption: A timestamp alone makes a record trustworthy
What really happens: A timestamp is useful, but it does not prove context or ownership - Common assumption: A shared spreadsheet is enough for traceability
What really happens: Shared files often break version control and context - Common assumption: A record is complete if the final form is filled in
What really happens: Completeness also means the change history and supporting evidence are intact - Common assumption: A digital file is automatically better than paper
What really happens: Digital records still need governance, retention, and review discipline
Where ALCOA+ breaks in practice
The most common failure is not one missing field. It is the loss of the record’s story.
- Attributable: People share logins or edit records without a clear owner
Why it matters: You cannot defend who did what - Complete: Supporting notes or follow-up evidence are missing
Why it matters: The record no longer tells the full story - Consistent: Information appears in different systems with mismatched timing
Why it matters: Reviewers lose confidence in the sequence - Enduring: Files are saved in unstable locations or overwritten
Why it matters: The record cannot be relied on later - Available: Records take too long to retrieve
Why it matters: Inspection and review become harder than they should be
A short comparison
- Updates happen across files and inboxes: Records stay in one controlled workflow
- Traceability is assembled after the fact: History is attached to the action
- Review depends on memory and follow-up: Review is visible in the system
- Gaps appear during audit prep: Evidence is created as work happens
This is the practical value of ALCOA+: it gives teams a simple way to check whether the record still makes sense after the work is done.
What the FDA materials emphasize
The FDA’s data-integrity guidance focuses on reliability, accuracy, and risk-based control. Recent FDA data-quality resources continue that theme by stressing that documentation should be attributable, legible, contemporaneous, original or a true copy, and accurate.
For pharma teams, the practical question is simple:
- Can we trust the source record?
- Can we reconstruct the activity later?
- Can we explain why the record changed?
- Can we retrieve it without rebuilding it from scattered files?
If the answer is no, ALCOA+ is not being met in a meaningful way.
What a better system changes
A controlled eQMS does not just store data more neatly.
It changes the way the record is formed:
- the action is captured in context
- approvals are attached to the workflow
- audit trails preserve the history
- record retention is controlled
- review does not depend on manual reconstruction
That is the difference between a file that holds information and a record that can be trusted.
Download template
If your team wants to assess data-integrity risk, a short checklist is more useful than a broad policy document.
The checklist should help teams ask:
- Who owns the record?
- Is the source data preserved?
- Can the history be reconstructed?
- Does the record remain available over time?
That gives QA and compliance teams a practical starting point before they look at system changes.
Complere fit
Complere is relevant here because ALCOA+ is ultimately about governed evidence, not just record storage.
The platform becomes useful when teams need to keep the record, the approval path, and the supporting evidence attached in one governed workflow instead of rebuilding the story from files, emails, and memory.
That matters when QA needs to prove not only that a record exists, but that it still holds together under review.
Closing thought
ALCOA+ is less about memorizing the letters and more about seeing whether a record still makes sense when someone else picks it up later.
If the record is trustworthy, the quality decision is easier to trust too.
If your current process still depends on stitching together evidence after the fact, the next step is a governed workflow that keeps the story intact from the start.
Disclaimer
This article is a practical interpretation of FDA data-integrity expectations and is not legal advice. Teams should assess ALCOA+ controls against their specific process, data type, and validation approach.


.jpeg)
.jpeg)
.jpeg)
