Download practical templates used by quality teams to investigate events, manage CAPA, assess change impact, evaluate training effectiveness, and strengthen compliance systems.
Use these resources to standardise quality work now, then connect the same processes inside Complere as your quality operations mature.
Practical templates used by regulated teams to investigate events, manage CAPA, assess compliance, and strengthen quality systems.
A step-by-step workflow outlining how deviation investigations progress from event reporting to CAPA implementation and closure.
A buyer-focused checklist for evaluating whether an electronic quality management system supports key 21 CFR Part 11 compliance expectations.
A toolkit containing practical worksheets for structured root cause investigations using 5 Whys and Fishbone analysis methods.
Evaluate the operational, quality, training, and regulatory impact of proposed changes before implementation.
A practical template for documenting deviations, investigation details, and immediate corrective actions in a structured GMP-compliant format.
A structured SOP template for managing corrective and preventive actions, including root cause investigation, action ownership, and effectiveness verification.
Preview one of the most-used quality templates before downloading. This example shows how deviation investigations can be documented with clear reporting fields, root cause analysis steps, and CAPA linkage for closure.
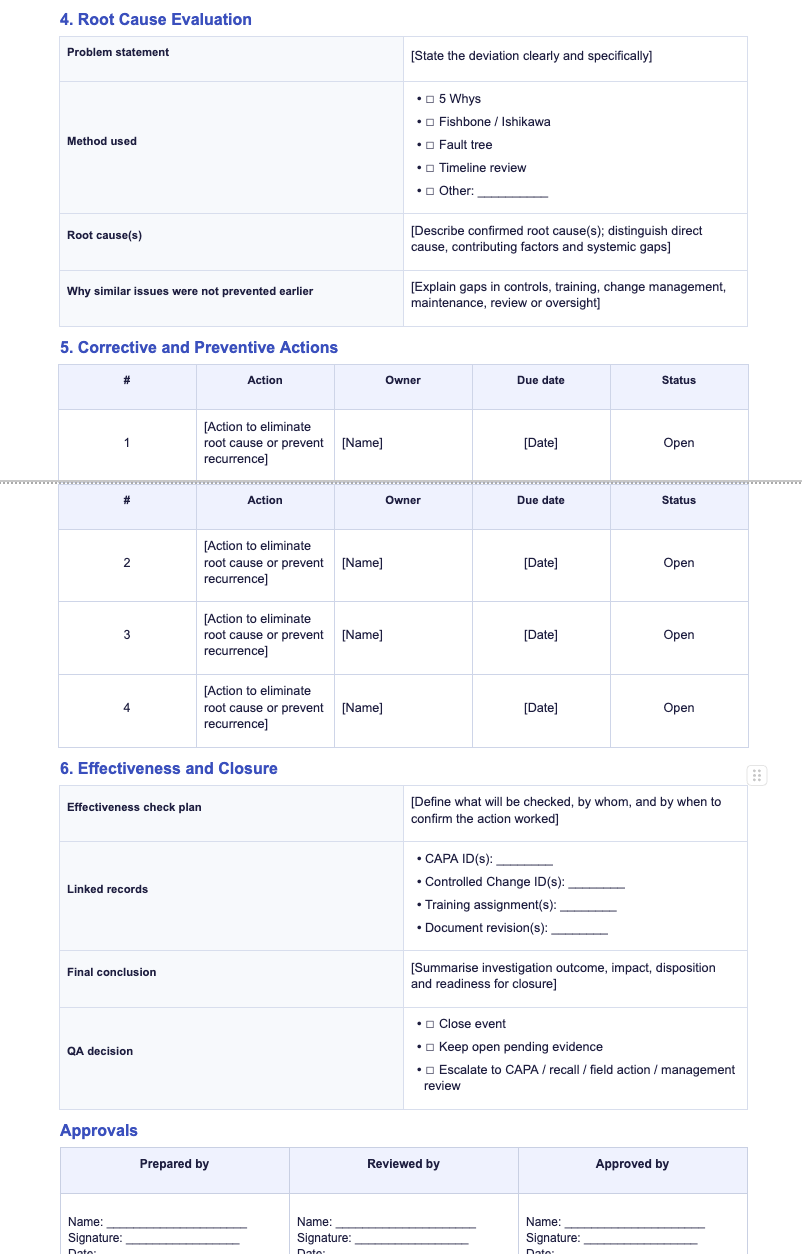
A practical template for documenting deviations, investigation details, and immediate corrective actions in a structured GMP-compliant format.



Whether you operate in pharmaceutical manufacturing, medical devices, healthcare, nutraceuticals, or contract manufacturing, Complere provides structured, connected, and inspection-ready quality systems tailored to your regulatory environment.
